ZITHROMAX® IV Description
(azithromycin dihydrate IV)
11 DESCRIPTION
ZITHROMAX for injection contains the active ingredient azithromycin, an azalide, a subclass of macrolide antibacterial drug, for intravenous injection. Azithromycin has the chemical name (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13- [(2,6-dideoxy-3-C-methyl-3-O -methyl-α-L-ribo-hexopyranosyl)oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-hepta-methyl- 11- [[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy]-1-oxa- 6-azacyclopentadecan-15-one. Azithromycin is derived from erythromycin; however, it differs chemically from erythromycin in that a methyl-substituted nitrogen atom is incorporated into the lactone ring. Its molecular formula is C38H72N2O12, and its molecular weight is 749. Azithromycin has the following structural formula:
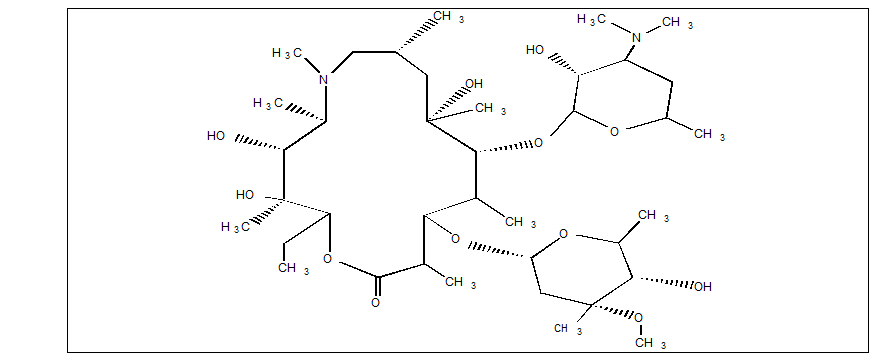
Azithromycin, as the dihydrate, is a white crystalline powder with a molecular formula of C38H72N2O12∙ 2H2O and a molecular weight of 785.
ZITHROMAX for injection consists of azithromycin dihydrate and the following inactive ingredients: anhydrous citric acid and sodium hydroxide. Sodium hydroxide is added to adjust the pH. ZITHROMAX for injection is supplied as white to off-white lyophilized powder in a single-dose vial for intravenous administration. Each vial contains azithromycin dihydrate equivalent to 500 mg of azithromycin, 384.6 mg anhydrous citric acid, and sodium hydroxide. Reconstitution, according to label directions, results in approximately 5 mL of ZITHROMAX for intravenous injection with each mL containing azithromycin dihydrate equivalent to 100 mg of azithromycin, 76.9 mg anhydrous citric acid, and sodium hydroxide.
Find ZITHROMAX® IV medical information:
Find ZITHROMAX® IV medical information:
ZITHROMAX® IV Quick Finder
Health Professional Information
Description
11 DESCRIPTION
ZITHROMAX for injection contains the active ingredient azithromycin, an azalide, a subclass of macrolide antibacterial drug, for intravenous injection. Azithromycin has the chemical name (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13- [(2,6-dideoxy-3-C-methyl-3-O -methyl-α-L-ribo-hexopyranosyl)oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-hepta-methyl- 11- [[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy]-1-oxa- 6-azacyclopentadecan-15-one. Azithromycin is derived from erythromycin; however, it differs chemically from erythromycin in that a methyl-substituted nitrogen atom is incorporated into the lactone ring. Its molecular formula is C38H72N2O12, and its molecular weight is 749. Azithromycin has the following structural formula:

Azithromycin, as the dihydrate, is a white crystalline powder with a molecular formula of C38H72N2O12∙ 2H2O and a molecular weight of 785.
ZITHROMAX for injection consists of azithromycin dihydrate and the following inactive ingredients: anhydrous citric acid and sodium hydroxide. Sodium hydroxide is added to adjust the pH. ZITHROMAX for injection is supplied as white to off-white lyophilized powder in a single-dose vial for intravenous administration. Each vial contains azithromycin dihydrate equivalent to 500 mg of azithromycin, 384.6 mg anhydrous citric acid, and sodium hydroxide. Reconstitution, according to label directions, results in approximately 5 mL of ZITHROMAX for intravenous injection with each mL containing azithromycin dihydrate equivalent to 100 mg of azithromycin, 76.9 mg anhydrous citric acid, and sodium hydroxide.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.