XELJANZ / XELJANZ XR Full Patient Information
(tofacitinib)
Full Patient Information
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: December 2021 | ||||
| MEDICATION GUIDE | |||||
| XELJANZ (ZEL' JANS') (tofacitinib) tablets, for oral use | XELJANZ XR (ZEL' JANS' EKS-AHR) (tofacitinib) extended-release tablets, for oral use | XELJANZ (ZEL' JANS') (tofacitinib) Oral Solution | |||
What is the most important information I should know about XELJANZ/XELJANZ XR/XELJANZ Oral Solution? | |||||
| 1. | Serious infections. XELJANZ/XELJANZ XR/XELJANZ Oral Solution is a medicine that affects your immune system. XELJANZ/XELJANZ XR/XELJANZ Oral Solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
People with ulcerative colitis taking the higher dose of XELJANZ (10 mg twice daily) or XELJANZ XR (22 mg one time each day) have a higher risk of serious infections and shingles. Before starting XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider if you:
| ||||
|
| ||||
| |||||
| 2. | Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. | ||||
| 3. | Cancer and immune system problems. XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase your risk of certain cancers by changing the way your immune system works.
| ||||
| 4. | Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily, especially if you are a current or past smoker. Get emergency help right away if you have any symptoms of a heart attack or stroke while taking XELJANZ, including:
| ||||
| 5. | Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. Blood clots in the lungs have also happened in people with ulcerative colitis. Some people have died from these blood clots.
| ||||
| 6. | Tears (perforation) in the stomach or intestines.
| ||||
| 7. | Allergic reactions.
| ||||
| 8. | Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and while you take XELJANZ/XELJANZ XR/XELJANZ Oral Solution to check for the following side effects:
You should not take XELJANZ/XELJANZ XR/XELJANZ Oral Solution if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high. Your healthcare provider may stop your XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment for a period of time if needed because of changes in these blood test results. You may also have changes in other laboratory tests, such as your blood cholesterol levels. Your healthcare provider should do blood tests to check your cholesterol levels 4 to 8 weeks after you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, and as needed after that. Normal cholesterol levels are important to good heart health. | ||||
| See "What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution?" for more information about side effects. | |||||
What is XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
XELJANZ/XELJANZ XR/XELJANZ Oral Solution is not recommended for people with severe liver problems. It is not known if XELJANZ/XELJANZ Oral Solution is safe and effective in children for treatment other than active polyarticular course juvenile arthritis. It is not known if XELJANZ XR is safe and effective in children. | |||||
| What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
| |||||
| How should I take XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Take XELJANZ/XELJANZ XR/XELJANZ Oral Solution exactly as your healthcare provider tells you to take it.
| |||||
What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution? XELJANZ/XELJANZ XR/XELJANZ Oral Solution may cause serious side effects, including:
| |||||
|
| ||||
Common side effects of XELJANZ/XELJANZ XR in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
These are not all the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Pfizer at 1-800-438-1985. | |||||
How should I store XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
| |||||
| General information about the safe and effective use of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XELJANZ/XELJANZ XR/XELJANZ Oral Solution for a condition for which it was not prescribed. Do not give XELJANZ/XELJANZ XR/XELJANZ Oral Solution to other people, even if they have the same symptoms you have. It may harm them. This Medication Guide summarizes the most important information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution that is written for health professionals. | |||||
| What are the ingredients in XELJANZ 5 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ 10 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ XR 11 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, and triacetin. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ XR 22 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ Oral Solution? Active ingredient: tofacitinib citrate Inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
LAB-0535-13.0 | |||||
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Serious Infections
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may lower the ability of their immune system to fight infections. Advise patients not to start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution if they have an active infection. Instruct patients to contact their healthcare provider immediately during treatment if symptoms suggesting infection appear in order to ensure rapid evaluation and appropriate treatment [see Warnings and Precautions (5.1)].
Advise patients that the risk of herpes zoster, some cases of which can be serious, is increased in patients treated with XELJANZ/XELJANZ XR [see Warnings and Precautions (5.1)].
Malignancies and Lymphoproliferative Disorders
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of certain cancers, and that lymphoma and other cancers have been observed in patients taking XELJANZ. Instruct patients to inform their healthcare provider if they have ever had any type of cancer [see Warnings and Precautions (5.3)].
Major Adverse Cardiovascular Events
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of major adverse cardiovascular events (MACE) defined as myocardial infarction, stroke, and cardiovascular death. Instruct all patients, especially current or past smokers or patients with other cardiovascular risk factors, to be alert for the development of signs and symptoms of cardiovascular events [see Warnings and Precautions (5.4)].
Thrombosis
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of thrombosis (sudden shortness of breath, chest pain worsened with breathing, swelling of leg or arm, leg pain or tenderness, red or discolored skin in the affected leg or arm) [see Warnings and Precautions (5.5)].
Hypersensitivity
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of allergic reactions while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution [see Warnings and Precautions (5.7)].
Important Information on Laboratory Abnormalities
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may affect certain lab test results, and that blood tests are required before and during XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment [see Warnings and Precautions (5.8)].
Pregnancy
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females to inform their prescriber of a known or suspected pregnancy. Inform patients that Pfizer has a registry for pregnant women who have taken XELJANZ/XELJANZ XR/XELJANZ Oral Solution during pregnancy. Advise patients to contact the registry at 1-877-311-8972 to enroll [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed during treatment with XELJANZ/XELJANZ XR/XELJANZ Oral Solution and for at least 18 hours after the last dose of XELJANZ/XELJANZ Oral Solution or 36 hours after the last dose of XELJANZ XR [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)]. It is not known if this effect is reversible.
INSTRUCTIONS FOR USE
XELJANZ (ZEL' JANS')
(tofacitinib)
Oral Solution
Read this Instructions for Use before you start taking XELJANZ Oral Solution and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring XELJANZ Oral Solution:
Always use the oral dosing syringe that comes with XELJANZ Oral Solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store XELJANZ?
- Store XELJANZ Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C).
- Always store XELJANZ Oral Solution in the original bottle and carton to protect from light.
Keep XELJANZ and all medicines out of the reach of children.
Use XELJANZ Oral Solution within 60 days of opening the bottle. Throw away (discard) remaining XELJANZ Oral Solution after 60 days.
To help you remember when to throw away your bottle of XELJANZ Oral Solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
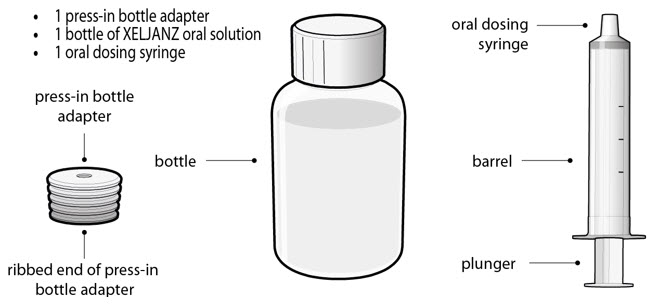
Each carton of XELJANZ Oral Solution contains:
- 1 press-in bottle adapter
- 1 bottle of XELJANZ Oral Solution
- 1 oral dosing syringe
Step 1. Remove bottle from carton
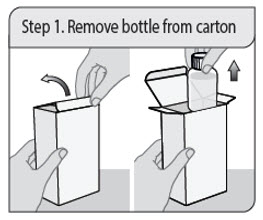
Open the carton and remove the bottle of XELJANZ Oral Solution.
Step 2. Open bottle
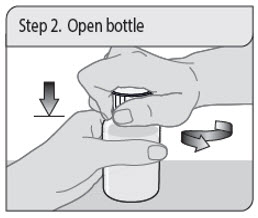
Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)
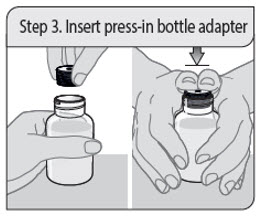
Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe
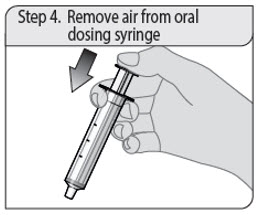
Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe
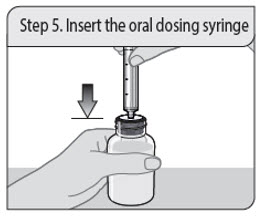
Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle
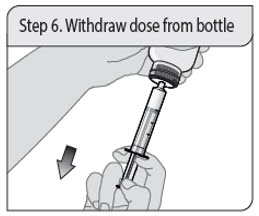
With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe
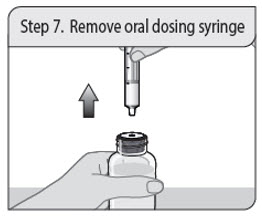
Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose
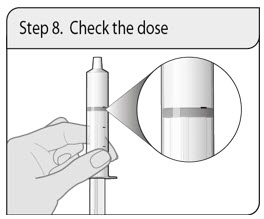
Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of XELJANZ
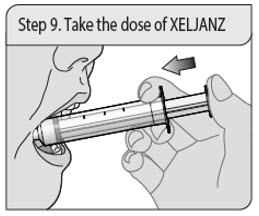
Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle
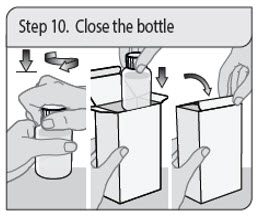
Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect XELJANZ Oral Solution from light.
Step 11. Clean oral dosing syringe
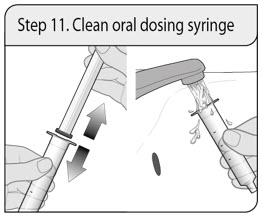
Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the XELJANZ Oral Solution.
Do not throw away the oral dosing syringe.

LAB-1422-1.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: September 2020
Find XELJANZ / XELJANZ XR medical information:
Find XELJANZ / XELJANZ XR medical information:
XELJANZ / XELJANZ XR Quick Finder
Health Professional Information
Full Patient Information
Full Patient Information
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: December 2021 | ||||
| MEDICATION GUIDE | |||||
| XELJANZ (ZEL' JANS') (tofacitinib) tablets, for oral use | XELJANZ XR (ZEL' JANS' EKS-AHR) (tofacitinib) extended-release tablets, for oral use | XELJANZ (ZEL' JANS') (tofacitinib) Oral Solution | |||
What is the most important information I should know about XELJANZ/XELJANZ XR/XELJANZ Oral Solution? | |||||
| 1. | Serious infections. XELJANZ/XELJANZ XR/XELJANZ Oral Solution is a medicine that affects your immune system. XELJANZ/XELJANZ XR/XELJANZ Oral Solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
People with ulcerative colitis taking the higher dose of XELJANZ (10 mg twice daily) or XELJANZ XR (22 mg one time each day) have a higher risk of serious infections and shingles. Before starting XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider if you:
| ||||
|
| ||||
| |||||
| 2. | Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. | ||||
| 3. | Cancer and immune system problems. XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase your risk of certain cancers by changing the way your immune system works.
| ||||
| 4. | Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily, especially if you are a current or past smoker. Get emergency help right away if you have any symptoms of a heart attack or stroke while taking XELJANZ, including:
| ||||
| 5. | Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. Blood clots in the lungs have also happened in people with ulcerative colitis. Some people have died from these blood clots.
| ||||
| 6. | Tears (perforation) in the stomach or intestines.
| ||||
| 7. | Allergic reactions.
| ||||
| 8. | Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and while you take XELJANZ/XELJANZ XR/XELJANZ Oral Solution to check for the following side effects:
You should not take XELJANZ/XELJANZ XR/XELJANZ Oral Solution if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high. Your healthcare provider may stop your XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment for a period of time if needed because of changes in these blood test results. You may also have changes in other laboratory tests, such as your blood cholesterol levels. Your healthcare provider should do blood tests to check your cholesterol levels 4 to 8 weeks after you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, and as needed after that. Normal cholesterol levels are important to good heart health. | ||||
| See "What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution?" for more information about side effects. | |||||
What is XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
XELJANZ/XELJANZ XR/XELJANZ Oral Solution is not recommended for people with severe liver problems. It is not known if XELJANZ/XELJANZ Oral Solution is safe and effective in children for treatment other than active polyarticular course juvenile arthritis. It is not known if XELJANZ XR is safe and effective in children. | |||||
| What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
| |||||
| How should I take XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Take XELJANZ/XELJANZ XR/XELJANZ Oral Solution exactly as your healthcare provider tells you to take it.
| |||||
What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution? XELJANZ/XELJANZ XR/XELJANZ Oral Solution may cause serious side effects, including:
| |||||
|
| ||||
Common side effects of XELJANZ/XELJANZ XR in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
These are not all the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Pfizer at 1-800-438-1985. | |||||
How should I store XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
| |||||
| General information about the safe and effective use of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XELJANZ/XELJANZ XR/XELJANZ Oral Solution for a condition for which it was not prescribed. Do not give XELJANZ/XELJANZ XR/XELJANZ Oral Solution to other people, even if they have the same symptoms you have. It may harm them. This Medication Guide summarizes the most important information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution that is written for health professionals. | |||||
| What are the ingredients in XELJANZ 5 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ 10 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ XR 11 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, and triacetin. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ XR 22 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ Oral Solution? Active ingredient: tofacitinib citrate Inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
LAB-0535-13.0 | |||||
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Serious Infections
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may lower the ability of their immune system to fight infections. Advise patients not to start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution if they have an active infection. Instruct patients to contact their healthcare provider immediately during treatment if symptoms suggesting infection appear in order to ensure rapid evaluation and appropriate treatment [see Warnings and Precautions (5.1)].
Advise patients that the risk of herpes zoster, some cases of which can be serious, is increased in patients treated with XELJANZ/XELJANZ XR [see Warnings and Precautions (5.1)].
Malignancies and Lymphoproliferative Disorders
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of certain cancers, and that lymphoma and other cancers have been observed in patients taking XELJANZ. Instruct patients to inform their healthcare provider if they have ever had any type of cancer [see Warnings and Precautions (5.3)].
Major Adverse Cardiovascular Events
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of major adverse cardiovascular events (MACE) defined as myocardial infarction, stroke, and cardiovascular death. Instruct all patients, especially current or past smokers or patients with other cardiovascular risk factors, to be alert for the development of signs and symptoms of cardiovascular events [see Warnings and Precautions (5.4)].
Thrombosis
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of thrombosis (sudden shortness of breath, chest pain worsened with breathing, swelling of leg or arm, leg pain or tenderness, red or discolored skin in the affected leg or arm) [see Warnings and Precautions (5.5)].
Hypersensitivity
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of allergic reactions while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution [see Warnings and Precautions (5.7)].
Important Information on Laboratory Abnormalities
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may affect certain lab test results, and that blood tests are required before and during XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment [see Warnings and Precautions (5.8)].
Pregnancy
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females to inform their prescriber of a known or suspected pregnancy. Inform patients that Pfizer has a registry for pregnant women who have taken XELJANZ/XELJANZ XR/XELJANZ Oral Solution during pregnancy. Advise patients to contact the registry at 1-877-311-8972 to enroll [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed during treatment with XELJANZ/XELJANZ XR/XELJANZ Oral Solution and for at least 18 hours after the last dose of XELJANZ/XELJANZ Oral Solution or 36 hours after the last dose of XELJANZ XR [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)]. It is not known if this effect is reversible.
INSTRUCTIONS FOR USE
XELJANZ (ZEL' JANS')
(tofacitinib)
Oral Solution
Read this Instructions for Use before you start taking XELJANZ Oral Solution and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring XELJANZ Oral Solution:
Always use the oral dosing syringe that comes with XELJANZ Oral Solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store XELJANZ?
- Store XELJANZ Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C).
- Always store XELJANZ Oral Solution in the original bottle and carton to protect from light.
Keep XELJANZ and all medicines out of the reach of children.
Use XELJANZ Oral Solution within 60 days of opening the bottle. Throw away (discard) remaining XELJANZ Oral Solution after 60 days.
To help you remember when to throw away your bottle of XELJANZ Oral Solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
Each carton of XELJANZ Oral Solution contains:
- 1 press-in bottle adapter
- 1 bottle of XELJANZ Oral Solution
- 1 oral dosing syringe

Step 1. Remove bottle from carton

Open the carton and remove the bottle of XELJANZ Oral Solution.
Step 2. Open bottle

Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)

Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe

Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe

Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle

With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe

Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose

Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of XELJANZ

Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle

Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect XELJANZ Oral Solution from light.
Step 11. Clean oral dosing syringe

Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the XELJANZ Oral Solution.
Do not throw away the oral dosing syringe.

LAB-1422-1.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: September 2020
Health Professional Information
Full Patient Information
Full Patient Information
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: December 2021 | ||||
| MEDICATION GUIDE | |||||
| XELJANZ (ZEL' JANS') (tofacitinib) tablets, for oral use | XELJANZ XR (ZEL' JANS' EKS-AHR) (tofacitinib) extended-release tablets, for oral use | XELJANZ (ZEL' JANS') (tofacitinib) Oral Solution | |||
What is the most important information I should know about XELJANZ/XELJANZ XR/XELJANZ Oral Solution? | |||||
| 1. | Serious infections. XELJANZ/XELJANZ XR/XELJANZ Oral Solution is a medicine that affects your immune system. XELJANZ/XELJANZ XR/XELJANZ Oral Solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
People with ulcerative colitis taking the higher dose of XELJANZ (10 mg twice daily) or XELJANZ XR (22 mg one time each day) have a higher risk of serious infections and shingles. Before starting XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider if you:
| ||||
|
| ||||
| |||||
| 2. | Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. | ||||
| 3. | Cancer and immune system problems. XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase your risk of certain cancers by changing the way your immune system works.
| ||||
| 4. | Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily, especially if you are a current or past smoker. Get emergency help right away if you have any symptoms of a heart attack or stroke while taking XELJANZ, including:
| ||||
| 5. | Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking XELJANZ 5 mg twice daily or XELJANZ 10 mg twice daily. Blood clots in the lungs have also happened in people with ulcerative colitis. Some people have died from these blood clots.
| ||||
| 6. | Tears (perforation) in the stomach or intestines.
| ||||
| 7. | Allergic reactions.
| ||||
| 8. | Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and while you take XELJANZ/XELJANZ XR/XELJANZ Oral Solution to check for the following side effects:
You should not take XELJANZ/XELJANZ XR/XELJANZ Oral Solution if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high. Your healthcare provider may stop your XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment for a period of time if needed because of changes in these blood test results. You may also have changes in other laboratory tests, such as your blood cholesterol levels. Your healthcare provider should do blood tests to check your cholesterol levels 4 to 8 weeks after you start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, and as needed after that. Normal cholesterol levels are important to good heart health. | ||||
| See "What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution?" for more information about side effects. | |||||
What is XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
XELJANZ/XELJANZ XR/XELJANZ Oral Solution is not recommended for people with severe liver problems. It is not known if XELJANZ/XELJANZ Oral Solution is safe and effective in children for treatment other than active polyarticular course juvenile arthritis. It is not known if XELJANZ XR is safe and effective in children. | |||||
| What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Before taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
| |||||
| How should I take XELJANZ/XELJANZ XR/XELJANZ Oral Solution? Take XELJANZ/XELJANZ XR/XELJANZ Oral Solution exactly as your healthcare provider tells you to take it.
| |||||
What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution? XELJANZ/XELJANZ XR/XELJANZ Oral Solution may cause serious side effects, including:
| |||||
|
| ||||
Common side effects of XELJANZ/XELJANZ XR in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
These are not all the possible side effects of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Pfizer at 1-800-438-1985. | |||||
How should I store XELJANZ/XELJANZ XR/XELJANZ Oral Solution?
| |||||
| General information about the safe and effective use of XELJANZ/XELJANZ XR/XELJANZ Oral Solution. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XELJANZ/XELJANZ XR/XELJANZ Oral Solution for a condition for which it was not prescribed. Do not give XELJANZ/XELJANZ XR/XELJANZ Oral Solution to other people, even if they have the same symptoms you have. It may harm them. This Medication Guide summarizes the most important information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about XELJANZ/XELJANZ XR/XELJANZ Oral Solution that is written for health professionals. | |||||
| What are the ingredients in XELJANZ 5 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ 10 mg? Active ingredient: tofacitinib citrate Inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin. What are the ingredients in XELJANZ XR 11 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, and triacetin. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ XR 22 mg? Active ingredient: tofacitinib citrate Inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze. What are the ingredients in XELJANZ Oral Solution? Active ingredient: tofacitinib citrate Inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
LAB-0535-13.0 | |||||
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Serious Infections
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may lower the ability of their immune system to fight infections. Advise patients not to start taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution if they have an active infection. Instruct patients to contact their healthcare provider immediately during treatment if symptoms suggesting infection appear in order to ensure rapid evaluation and appropriate treatment [see Warnings and Precautions (5.1)].
Advise patients that the risk of herpes zoster, some cases of which can be serious, is increased in patients treated with XELJANZ/XELJANZ XR [see Warnings and Precautions (5.1)].
Malignancies and Lymphoproliferative Disorders
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of certain cancers, and that lymphoma and other cancers have been observed in patients taking XELJANZ. Instruct patients to inform their healthcare provider if they have ever had any type of cancer [see Warnings and Precautions (5.3)].
Major Adverse Cardiovascular Events
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may increase their risk of major adverse cardiovascular events (MACE) defined as myocardial infarction, stroke, and cardiovascular death. Instruct all patients, especially current or past smokers or patients with other cardiovascular risk factors, to be alert for the development of signs and symptoms of cardiovascular events [see Warnings and Precautions (5.4)].
Thrombosis
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of thrombosis (sudden shortness of breath, chest pain worsened with breathing, swelling of leg or arm, leg pain or tenderness, red or discolored skin in the affected leg or arm) [see Warnings and Precautions (5.5)].
Hypersensitivity
Advise patients to stop taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution and to call their healthcare provider right away if they experience any symptoms of allergic reactions while taking XELJANZ/XELJANZ XR/XELJANZ Oral Solution [see Warnings and Precautions (5.7)].
Important Information on Laboratory Abnormalities
Inform patients that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may affect certain lab test results, and that blood tests are required before and during XELJANZ/XELJANZ XR/XELJANZ Oral Solution treatment [see Warnings and Precautions (5.8)].
Pregnancy
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females to inform their prescriber of a known or suspected pregnancy. Inform patients that Pfizer has a registry for pregnant women who have taken XELJANZ/XELJANZ XR/XELJANZ Oral Solution during pregnancy. Advise patients to contact the registry at 1-877-311-8972 to enroll [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed during treatment with XELJANZ/XELJANZ XR/XELJANZ Oral Solution and for at least 18 hours after the last dose of XELJANZ/XELJANZ Oral Solution or 36 hours after the last dose of XELJANZ XR [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential that XELJANZ/XELJANZ XR/XELJANZ Oral Solution may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)]. It is not known if this effect is reversible.
INSTRUCTIONS FOR USE
XELJANZ (ZEL' JANS')
(tofacitinib)
Oral Solution
Read this Instructions for Use before you start taking XELJANZ Oral Solution and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring XELJANZ Oral Solution:
Always use the oral dosing syringe that comes with XELJANZ Oral Solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store XELJANZ?
- Store XELJANZ Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C).
- Always store XELJANZ Oral Solution in the original bottle and carton to protect from light.
Keep XELJANZ and all medicines out of the reach of children.
Use XELJANZ Oral Solution within 60 days of opening the bottle. Throw away (discard) remaining XELJANZ Oral Solution after 60 days.
To help you remember when to throw away your bottle of XELJANZ Oral Solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
Each carton of XELJANZ Oral Solution contains:
- 1 press-in bottle adapter
- 1 bottle of XELJANZ Oral Solution
- 1 oral dosing syringe

Step 1. Remove bottle from carton

Open the carton and remove the bottle of XELJANZ Oral Solution.
Step 2. Open bottle

Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)

Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe

Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe

Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle

With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe

Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose

Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of XELJANZ

Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle

Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect XELJANZ Oral Solution from light.
Step 11. Clean oral dosing syringe

Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the XELJANZ Oral Solution.
Do not throw away the oral dosing syringe.

LAB-1422-1.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: September 2020
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Contact Medical Information.9AM-5PM ET Monday to Friday; excluding holidays.
Report Adverse Event
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.
