THROMBIN-JMI® Dosage and Administration
(thrombin, topical, bovine origin)
2 DOSAGE AND ADMINISTRATION
For topical use on the surface of bleeding tissue only. Do not inject.
2.1 Reconstitution
- •
- For routine use, reconstitute THROMBIN-JMI with sterile isotonic saline at a recommended concentration of 1,000 to 2,000 IU per mL.
- •
- Where bleeding is profuse, as from abraded surfaces of liver or spleen, concentrations of 1,000 IU per mL may be required.
- •
- For general use in plastic surgery, dental extractions, skin grafting, etc. solutions containing approximately 100 IU per mL are frequently used. Prepare intermediate strengths to suit the needs of the case by diluting the contents of the THROMBIN-JMI container with an appropriate volume of sterile isotonic saline. THROMBIN-JMI can be used in a dry form on oozing surfaces.
- •
- In instances where a concentration of approximately 1,000 IU per mL is desired, the contents of the vial of sterile isotonic saline diluent may be transferred into the THROMBIN-JMI container with a sterile syringe or sterile transfer device.
- •
- For use with GEL-FLOW™ NT, reconstitute THROMBIN-JMI 5,000 IU powder with 5 mL sterile isotonic saline and use THROMBIN-JMI thrombin solution according to the directions for use in the GEL-FLOW™ NT package insert.
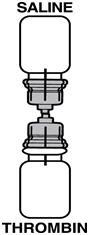
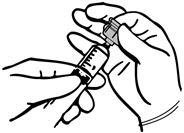
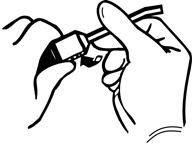
If the transfer device is used for reconstitution, transfer the diluent in the following manner:
- 1.
- Remove the plastic cap off of the diluent vial.
- 2.
- Remove the Tyvek cover from the transfer device container. Do not remove the device from the package.
- 3.
- Seat the blue end of the device on the diluent vial, pushing down until the spike penetrates the diaphragm and the device snaps in place.
- 4.
- Flip the plastic cover off on the THROMBIN-JMI container. DO NOT REMOVE THE ALUMINUM SEAL.
- 5.
- Remove the plastic package from the transfer device. Do not touch the exposed end of the device.
- 6.
- Invert the vial of diluent and insert the clear end of the transfer device into the diaphragm of the THROMBIN-JMI container.
CAUTION: Solutions should be used promptly upon removal from the container. However, the solution may be refrigerated at 2°C – 8°C for up to 24 hours, or may be stored at room temperature for up to 8 hours after reconstitution.
THROMBIN-JMI PUMP SPRAY KIT
Refer to THROMBIN-JMI Pump Spray Kit instructions for spray pump assembly and use.
Each spray kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray pump and actuator.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on inner tray to expose sterile contents.
- 3.
- Reconstitute the THROMBIN-JMI to desired potency by introducing sterile isotonic saline with a sterile syringe or a sterile transfer device. If the transfer device is used, follow the previously described procedure.
- 4.
- When the THROMBIN-JMI is completely dissolved, open vial by flipping up metal and tearing counterclockwise.
- 5.
- Remove the rubber diaphragm from vial. Remove pump with protective cap from tray and snap onto vial.
- 6.
- Remove protective cap and attach actuator.
- 7.
- To spray, hold vial upright or at a slight angle. Several strokes of the pump will be required to expel the solution.
- 8.
- Discard unused contents and pump: DO NOT TRANSFER SPRAY PUMP TO ANOTHER VIAL.
THROMBIN-JMI SYRINGE SPRAY KIT
Refer to THROMBIN-JMI Syringe Spray Kit instructions for spray syringe assembly and use. Each syringe kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray tip and syringe.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
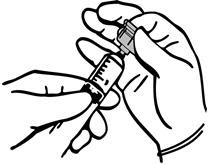
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix spray tip by pushing down and turning clockwise until the spray tip locks in place.
- 8.
- To spray, depress the syringe plunger in a normal fashion to dispense the THROMBIN-JMI thrombin solution through the tip in a fine spray.
- 9.
- Discard unused contents and syringe.
For use of THROMBIN-JMI 5,000 IU Syringe Spray Kit with GEL-FLOW™ NT:
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw all of the 5 mL of the saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- DISCARD the SPRAY TIP.
- 8.
- Use THROMBIN-JMI thrombin solution syringe according to the directions for use in the GEL-FLOW™ NT package insert.
THROMBIN-JMI EPISTAXIS KIT
Refer to THROMBIN-JMI Epistaxis Kit instructions for accessory assembly and use.
Each epistaxis kit contains one vial of THROMBIN-JMI, one vial of diluent and one nasal drug delivery device.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix the nasal drug delivery device on to the syringe by pushing the device down onto the THROMBIN-JMI thrombin solution filled syringe and turn clockwise until the nasal drug delivery device locks in place.
- 8.
- Insert the nasal drug delivery device into the naris and spray the THROMBIN-JMI thrombin solution onto the nasal mucosa through the nasal drug delivery device by depressing the syringe plunger using mild or moderate pressure on the syringe plunger.
- 9.
- After administration of THROMBIN-JMI, the device may be removed immediately or briefly held in the nasal passage.
- 10.
- Discard the unused contents, nasal drug delivery device, and the syringe.
2.2 Administration
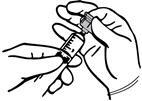
Topical application of THROMBIN-JMI
- 1.
- The recipient surface should be sponged (not wiped) free of blood before THROMBIN-JMI is applied.
- 2.
- A spray may be used or the surface may be flooded using a sterile syringe and small gauge needle. The most effective hemostasis results occur when the THROMBIN-JMI mixes freely with the blood as soon as it reaches the surface.
- 3.
- Sponging of the treated surfaces should be avoided to assure that the clot remains securely in place.
Use in conjunction with Absorbable Gelatin Sponge
Consult the Absorbable Gelatin Sponge, USP labeling for complete information for use prior to utilizing the following thrombin saturated sponge procedure.
- 1.
- Prepare THROMBIN-JMI solution to desired strength.
- 2.
- Immerse sponge strips of the desired size in THROMBIN-JMI solution. Knead the sponge strips vigorously with moistened, gloved fingers to remove trapped air, thereby facilitating saturation of the sponge.
- 3.
- Apply saturated sponge to bleeding area. Hold in place with a pledget of cotton or a small gauze sponge until hemostasis occurs.
Find THROMBIN-JMI® medical information:
Find THROMBIN-JMI® medical information:
THROMBIN-JMI® Quick Finder
Health Professional Information
Dosage and Administration
2 DOSAGE AND ADMINISTRATION
For topical use on the surface of bleeding tissue only. Do not inject.
2.1 Reconstitution
- •
- For routine use, reconstitute THROMBIN-JMI with sterile isotonic saline at a recommended concentration of 1,000 to 2,000 IU per mL.
- •
- Where bleeding is profuse, as from abraded surfaces of liver or spleen, concentrations of 1,000 IU per mL may be required.
- •
- For general use in plastic surgery, dental extractions, skin grafting, etc. solutions containing approximately 100 IU per mL are frequently used. Prepare intermediate strengths to suit the needs of the case by diluting the contents of the THROMBIN-JMI container with an appropriate volume of sterile isotonic saline. THROMBIN-JMI can be used in a dry form on oozing surfaces.
- •
- In instances where a concentration of approximately 1,000 IU per mL is desired, the contents of the vial of sterile isotonic saline diluent may be transferred into the THROMBIN-JMI container with a sterile syringe or sterile transfer device.
- •
- For use with GEL-FLOW™ NT, reconstitute THROMBIN-JMI 5,000 IU powder with 5 mL sterile isotonic saline and use THROMBIN-JMI thrombin solution according to the directions for use in the GEL-FLOW™ NT package insert.
If the transfer device is used for reconstitution, transfer the diluent in the following manner:
- 1.
- Remove the plastic cap off of the diluent vial.
- 2.
- Remove the Tyvek cover from the transfer device container. Do not remove the device from the package.
- 3.
- Seat the blue end of the device on the diluent vial, pushing down until the spike penetrates the diaphragm and the device snaps in place.
- 4.
- Flip the plastic cover off on the THROMBIN-JMI container. DO NOT REMOVE THE ALUMINUM SEAL.
- 5.
- Remove the plastic package from the transfer device. Do not touch the exposed end of the device.
- 6.
- Invert the vial of diluent and insert the clear end of the transfer device into the diaphragm of the THROMBIN-JMI container.
CAUTION: Solutions should be used promptly upon removal from the container. However, the solution may be refrigerated at 2°C – 8°C for up to 24 hours, or may be stored at room temperature for up to 8 hours after reconstitution.
THROMBIN-JMI PUMP SPRAY KIT
Refer to THROMBIN-JMI Pump Spray Kit instructions for spray pump assembly and use.
Each spray kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray pump and actuator.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on inner tray to expose sterile contents.
- 3.
- Reconstitute the THROMBIN-JMI to desired potency by introducing sterile isotonic saline with a sterile syringe or a sterile transfer device. If the transfer device is used, follow the previously described procedure.
- 4.
- When the THROMBIN-JMI is completely dissolved, open vial by flipping up metal and tearing counterclockwise.
- 5.
- Remove the rubber diaphragm from vial. Remove pump with protective cap from tray and snap onto vial.
- 6.
- Remove protective cap and attach actuator.
- 7.
- To spray, hold vial upright or at a slight angle. Several strokes of the pump will be required to expel the solution.
- 8.
- Discard unused contents and pump: DO NOT TRANSFER SPRAY PUMP TO ANOTHER VIAL.
THROMBIN-JMI SYRINGE SPRAY KIT
Refer to THROMBIN-JMI Syringe Spray Kit instructions for spray syringe assembly and use. Each syringe kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray tip and syringe.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix spray tip by pushing down and turning clockwise until the spray tip locks in place.
- 8.
- To spray, depress the syringe plunger in a normal fashion to dispense the THROMBIN-JMI thrombin solution through the tip in a fine spray.
- 9.
- Discard unused contents and syringe.
For use of THROMBIN-JMI 5,000 IU Syringe Spray Kit with GEL-FLOW™ NT:
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw all of the 5 mL of the saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- DISCARD the SPRAY TIP.
- 8.
- Use THROMBIN-JMI thrombin solution syringe according to the directions for use in the GEL-FLOW™ NT package insert.
THROMBIN-JMI EPISTAXIS KIT
Refer to THROMBIN-JMI Epistaxis Kit instructions for accessory assembly and use.
Each epistaxis kit contains one vial of THROMBIN-JMI, one vial of diluent and one nasal drug delivery device.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix the nasal drug delivery device on to the syringe by pushing the device down onto the THROMBIN-JMI thrombin solution filled syringe and turn clockwise until the nasal drug delivery device locks in place.
- 8.
- Insert the nasal drug delivery device into the naris and spray the THROMBIN-JMI thrombin solution onto the nasal mucosa through the nasal drug delivery device by depressing the syringe plunger using mild or moderate pressure on the syringe plunger.
- 9.
- After administration of THROMBIN-JMI, the device may be removed immediately or briefly held in the nasal passage.
- 10.
- Discard the unused contents, nasal drug delivery device, and the syringe.
2.2 Administration
Topical application of THROMBIN-JMI
- 1.
- The recipient surface should be sponged (not wiped) free of blood before THROMBIN-JMI is applied.
- 2.
- A spray may be used or the surface may be flooded using a sterile syringe and small gauge needle. The most effective hemostasis results occur when the THROMBIN-JMI mixes freely with the blood as soon as it reaches the surface.
- 3.
- Sponging of the treated surfaces should be avoided to assure that the clot remains securely in place.
Use in conjunction with Absorbable Gelatin Sponge
Consult the Absorbable Gelatin Sponge, USP labeling for complete information for use prior to utilizing the following thrombin saturated sponge procedure.
- 1.
- Prepare THROMBIN-JMI solution to desired strength.
- 2.
- Immerse sponge strips of the desired size in THROMBIN-JMI solution. Knead the sponge strips vigorously with moistened, gloved fingers to remove trapped air, thereby facilitating saturation of the sponge.
- 3.
- Apply saturated sponge to bleeding area. Hold in place with a pledget of cotton or a small gauze sponge until hemostasis occurs.
Health Professional Information
Dosage and Administration
2 DOSAGE AND ADMINISTRATION
For topical use on the surface of bleeding tissue only. Do not inject.
2.1 Reconstitution
- •
- For routine use, reconstitute THROMBIN-JMI with sterile isotonic saline at a recommended concentration of 1,000 to 2,000 IU per mL.
- •
- Where bleeding is profuse, as from abraded surfaces of liver or spleen, concentrations of 1,000 IU per mL may be required.
- •
- For general use in plastic surgery, dental extractions, skin grafting, etc. solutions containing approximately 100 IU per mL are frequently used. Prepare intermediate strengths to suit the needs of the case by diluting the contents of the THROMBIN-JMI container with an appropriate volume of sterile isotonic saline. THROMBIN-JMI can be used in a dry form on oozing surfaces.
- •
- In instances where a concentration of approximately 1,000 IU per mL is desired, the contents of the vial of sterile isotonic saline diluent may be transferred into the THROMBIN-JMI container with a sterile syringe or sterile transfer device.
- •
- For use with GEL-FLOW™ NT, reconstitute THROMBIN-JMI 5,000 IU powder with 5 mL sterile isotonic saline and use THROMBIN-JMI thrombin solution according to the directions for use in the GEL-FLOW™ NT package insert.
If the transfer device is used for reconstitution, transfer the diluent in the following manner:
- 1.
- Remove the plastic cap off of the diluent vial.
- 2.
- Remove the Tyvek cover from the transfer device container. Do not remove the device from the package.
- 3.
- Seat the blue end of the device on the diluent vial, pushing down until the spike penetrates the diaphragm and the device snaps in place.
- 4.
- Flip the plastic cover off on the THROMBIN-JMI container. DO NOT REMOVE THE ALUMINUM SEAL.
- 5.
- Remove the plastic package from the transfer device. Do not touch the exposed end of the device.
- 6.
- Invert the vial of diluent and insert the clear end of the transfer device into the diaphragm of the THROMBIN-JMI container.
CAUTION: Solutions should be used promptly upon removal from the container. However, the solution may be refrigerated at 2°C – 8°C for up to 24 hours, or may be stored at room temperature for up to 8 hours after reconstitution.
THROMBIN-JMI PUMP SPRAY KIT
Refer to THROMBIN-JMI Pump Spray Kit instructions for spray pump assembly and use.
Each spray kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray pump and actuator.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on inner tray to expose sterile contents.
- 3.
- Reconstitute the THROMBIN-JMI to desired potency by introducing sterile isotonic saline with a sterile syringe or a sterile transfer device. If the transfer device is used, follow the previously described procedure.
- 4.
- When the THROMBIN-JMI is completely dissolved, open vial by flipping up metal and tearing counterclockwise.
- 5.
- Remove the rubber diaphragm from vial. Remove pump with protective cap from tray and snap onto vial.
- 6.
- Remove protective cap and attach actuator.
- 7.
- To spray, hold vial upright or at a slight angle. Several strokes of the pump will be required to expel the solution.
- 8.
- Discard unused contents and pump: DO NOT TRANSFER SPRAY PUMP TO ANOTHER VIAL.
THROMBIN-JMI SYRINGE SPRAY KIT
Refer to THROMBIN-JMI Syringe Spray Kit instructions for spray syringe assembly and use. Each syringe kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray tip and syringe.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix spray tip by pushing down and turning clockwise until the spray tip locks in place.
- 8.
- To spray, depress the syringe plunger in a normal fashion to dispense the THROMBIN-JMI thrombin solution through the tip in a fine spray.
- 9.
- Discard unused contents and syringe.
For use of THROMBIN-JMI 5,000 IU Syringe Spray Kit with GEL-FLOW™ NT:
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw all of the 5 mL of the saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- DISCARD the SPRAY TIP.
- 8.
- Use THROMBIN-JMI thrombin solution syringe according to the directions for use in the GEL-FLOW™ NT package insert.
THROMBIN-JMI EPISTAXIS KIT
Refer to THROMBIN-JMI Epistaxis Kit instructions for accessory assembly and use.
Each epistaxis kit contains one vial of THROMBIN-JMI, one vial of diluent and one nasal drug delivery device.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix the nasal drug delivery device on to the syringe by pushing the device down onto the THROMBIN-JMI thrombin solution filled syringe and turn clockwise until the nasal drug delivery device locks in place.
- 8.
- Insert the nasal drug delivery device into the naris and spray the THROMBIN-JMI thrombin solution onto the nasal mucosa through the nasal drug delivery device by depressing the syringe plunger using mild or moderate pressure on the syringe plunger.
- 9.
- After administration of THROMBIN-JMI, the device may be removed immediately or briefly held in the nasal passage.
- 10.
- Discard the unused contents, nasal drug delivery device, and the syringe.
2.2 Administration
Topical application of THROMBIN-JMI
- 1.
- The recipient surface should be sponged (not wiped) free of blood before THROMBIN-JMI is applied.
- 2.
- A spray may be used or the surface may be flooded using a sterile syringe and small gauge needle. The most effective hemostasis results occur when the THROMBIN-JMI mixes freely with the blood as soon as it reaches the surface.
- 3.
- Sponging of the treated surfaces should be avoided to assure that the clot remains securely in place.
Use in conjunction with Absorbable Gelatin Sponge
Consult the Absorbable Gelatin Sponge, USP labeling for complete information for use prior to utilizing the following thrombin saturated sponge procedure.
- 1.
- Prepare THROMBIN-JMI solution to desired strength.
- 2.
- Immerse sponge strips of the desired size in THROMBIN-JMI solution. Knead the sponge strips vigorously with moistened, gloved fingers to remove trapped air, thereby facilitating saturation of the sponge.
- 3.
- Apply saturated sponge to bleeding area. Hold in place with a pledget of cotton or a small gauze sponge until hemostasis occurs.
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Contact Medical Information.9AM-5PM ET Monday to Friday; excluding holidays.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.