NURTEC® ODT Clinical Studies
(rimegepant)
14 CLINICAL STUDIES
14.1 Acute Treatment of Migraine
The efficacy of NURTEC ODT for the acute treatment of migraine with and without aura in adults was demonstrated in a randomized, double-blind, placebo-controlled trial: Study 1 (NCT03461757). Patients in the study were randomized to receive 75 mg of NURTEC ODT (N=732) or placebo (N=734). Patients were instructed to treat a migraine of moderate to severe headache pain intensity. Rescue medication (i.e., NSAIDs, acetaminophen, and/or an antiemetic) was allowed 2 hours after the initial treatment. Other forms of rescue medication such as triptans were not allowed within 48 hours of initial treatment. Approximately 14% of patients were taking preventive medications for migraine at baseline. None of the patients in Study 1 were on concomitant preventive medication that act on the CGRP pathway.
The primary efficacy analyses were conducted in patients who treated a migraine with moderate to severe pain. NURTEC ODT 75 mg demonstrated an effect on pain freedom and most bothersome symptom (MBS) freedom at two hours after dosing, compared to placebo. Pain freedom was defined as a reduction of moderate or severe headache pain to no headache pain, and MBS freedom was defined as the absence of the self-identified MBS (i.e., photophobia, phonophobia, or nausea). Among patients who selected an MBS, the most commonly selected symptom was photophobia (54%), followed by nausea (28%), and phonophobia (15%).
In Study 1, the percentage of patients achieving headache pain freedom and MBS freedom two hours after a single dose was statistically significantly greater in patients who received NURTEC ODT compared to those who received placebo (Table 1).
| Study 1 | ||
|---|---|---|
| NURTEC ODT 75 mg | Placebo | |
| ||
Pain Free at 2 hours | ||
n/N* | 142/669 | 74/682 |
% Responders | 21.2 | 10.9 |
Difference from placebo (%) | 10.3 | |
p-value | <0.001 | |
MBS Free at 2 hours | ||
n/N* | 235/669 | 183/682 |
% Responders | 35.1 | 26.8 |
Difference from placebo (%) | 8.3 | |
p-value | 0.001 | |
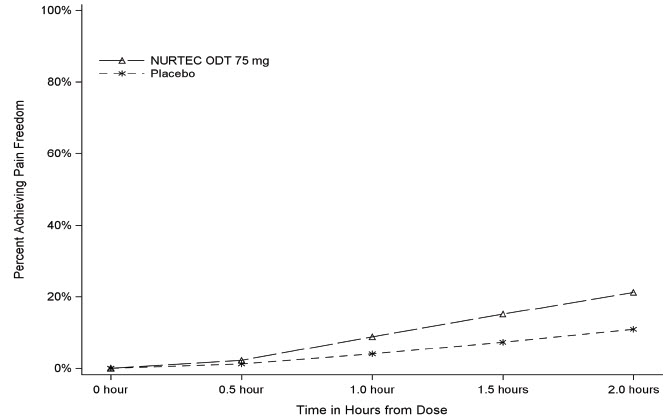
Figure 1 presents the percentage of patients achieving migraine pain freedom within 2 hours following treatment in Study 1.
Figure 1: Percentage of Patients Achieving Pain Freedom within 2 Hours in Study 1
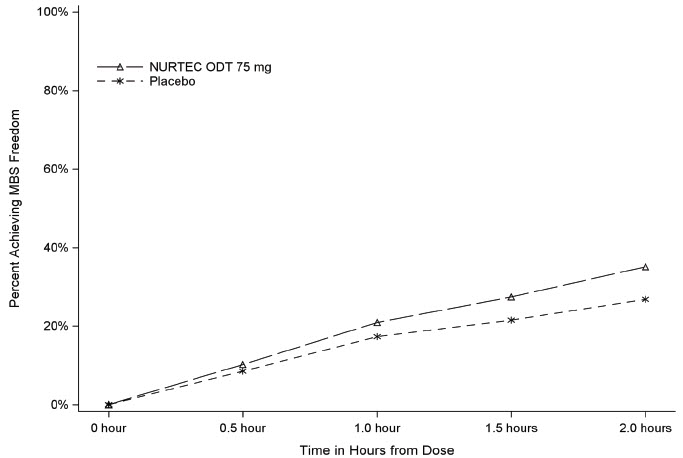
Figure 2 presents the percentage of patients achieving MBS freedom within 2 hours in Study 1.
Figure 2: Percentage of Patients Achieving MBS Freedom within 2 Hours in Study 1
In Study 1, statistically significant effects of NURTEC ODT compared to placebo were demonstrated for the additional efficacy endpoints of pain relief at 2 hours, sustained pain freedom 2-48 hours, use of rescue medication within 24 hours, and the percentage of patients reporting normal function at two hours after dosing (Table 2). Pain relief was defined as a reduction in migraine pain from moderate or severe severity to mild or none. The measurement of the percentage of patients reporting normal function at two hours after dosing was derived from a single item questionnaire, asking patients to select one response on a 4-point scale; normal function, mild impairment, severe impairment, or required bedrest.
| Study 1 | ||
|---|---|---|
| NURTEC ODT 75 mg | Placebo | |
Pain Relief at 2 hours | ||
n/N* | 397/669 | 295/682 |
% Responders | 59.3 | 43.3 |
Difference from placebo | 16.1 | |
p-value | <0.001 | |
Sustained Pain Freedom 2-48 hours | ||
n/N* | 90/669 | 37/682 |
% Responders | 13.5 | 5.4 |
Difference from placebo | 8.0 | |
p-value | <0.001 | |
Use of Rescue Medication within 24 hours† | ||
n/N* | 95/669 | 199/682 |
% Responders | 14.2 | 29.2 |
Difference from placebo | -15.0 | |
p-value | <0.001 | |
Percentage of Patients Reporting Normal Function at 2 hours | ||
n/N* | 255/669 | 176/682 |
% Responders | 38.1 | 25.8 |
Difference from placebo | 12.3 | |
p-value | <0.001 | |
The incidence of photophobia and phonophobia was reduced following administration of NURTEC ODT 75 mg as compared to placebo.
14.2 Preventive Treatment of Episodic Migraine
The efficacy of NURTEC ODT for the preventive treatment of episodic migraine in adults was demonstrated in one randomized, double-blind, placebo-controlled trial of a different oral dosage form of rimegepant (Study 2; NCT03732638).
Study 2 enrolled adult patients with at least a 1-year history of migraine (with or without aura). Patients experienced an average of 10.9 headache days during the 28-day observational period, which included an average of 10.2 migraine days, prior to randomization into the trial. Patients were randomized to receive every other day dosing of rimegepant 75 mg (N=373) or placebo (N=374) for 12 weeks. Patients were allowed to use acute headache treatments (i.e., triptans, NSAIDs, acetaminophen, antiemetics, muscle relaxants, and aspirin) as needed. Approximately 10% of patients were taking one preventive medication for migraine at baseline. The use of a concomitant medication that acts on the CGRP pathway was not permitted for either the acute or preventive treatment of migraine.
The study excluded patients with myocardial infarction, acute coronary syndrome, percutaneous coronary intervention, cardiac surgery, stroke, or transient ischemic attack within six months of screening.
The primary efficacy endpoint for Study 2 was the change from baseline in the mean number of monthly migraine days (MMDs) during Weeks 9 through 12 of the double-blind treatment phase.
The percentage of patients who achieved at least a 50% reduction from baseline in moderate to severe MMDs during Weeks 9 through 12 of the double-blind treatment phase compared to placebo was also evaluated. Rimegepant 75 mg dosed every other day demonstrated statistically significant improvements for these efficacy endpoints compared to placebo, as summarized in Table 3.
| Rimegepant 75 mg Every Other Day | Placebo Every Other Day | |
|---|---|---|
Monthly Migraine Days (MMD), Weeks 9-12 | N=348 | N=347 |
Change from baseline | -4.3 | -3.5 |
Change from placebo | -0.8 | |
p-value | 0.010 | |
≥ 50% Responders (Moderate to Severe MMDs), Weeks 9-12 | N=348 | N=347 |
% Responders | 49.1 | 41.5 |
Difference from placebo | 7.6 | |
p-value | 0.044 |
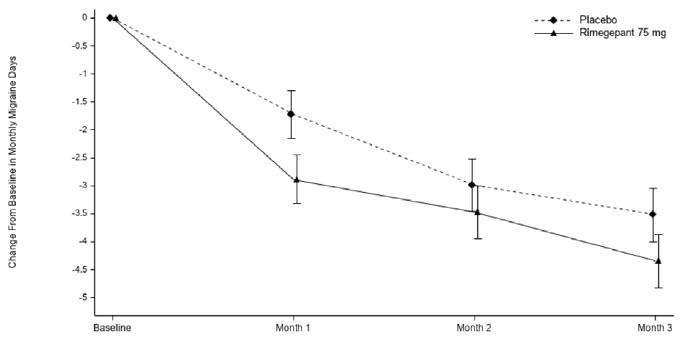
Figure 3: Change from Baseline in Monthly Migraine Days in Study 2a
aLeast-square means and 95% confidence intervals are presented.
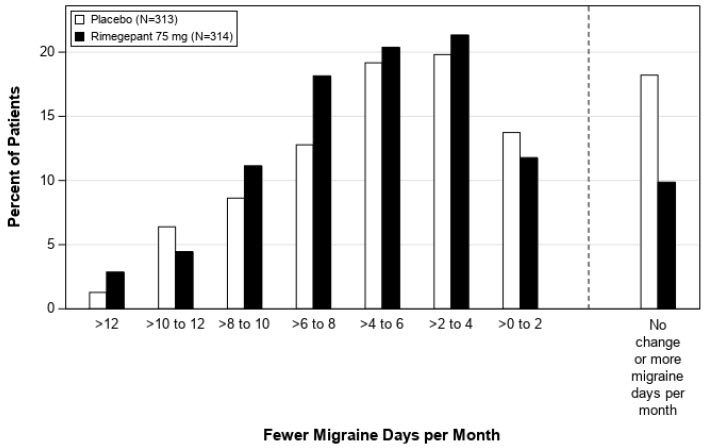
Figure 4: Distribution of Change from Baseline in Mean Monthly Migraine Days at Month 3 by Treatment Group in Study 2a
aFigure excludes patients with missing data.
Find NURTEC® ODT medical information:
Find NURTEC® ODT medical information:
NURTEC® ODT Quick Finder
Health Professional Information
Clinical Studies
14 CLINICAL STUDIES
14.1 Acute Treatment of Migraine
The efficacy of NURTEC ODT for the acute treatment of migraine with and without aura in adults was demonstrated in a randomized, double-blind, placebo-controlled trial: Study 1 (NCT03461757). Patients in the study were randomized to receive 75 mg of NURTEC ODT (N=732) or placebo (N=734). Patients were instructed to treat a migraine of moderate to severe headache pain intensity. Rescue medication (i.e., NSAIDs, acetaminophen, and/or an antiemetic) was allowed 2 hours after the initial treatment. Other forms of rescue medication such as triptans were not allowed within 48 hours of initial treatment. Approximately 14% of patients were taking preventive medications for migraine at baseline. None of the patients in Study 1 were on concomitant preventive medication that act on the CGRP pathway.
The primary efficacy analyses were conducted in patients who treated a migraine with moderate to severe pain. NURTEC ODT 75 mg demonstrated an effect on pain freedom and most bothersome symptom (MBS) freedom at two hours after dosing, compared to placebo. Pain freedom was defined as a reduction of moderate or severe headache pain to no headache pain, and MBS freedom was defined as the absence of the self-identified MBS (i.e., photophobia, phonophobia, or nausea). Among patients who selected an MBS, the most commonly selected symptom was photophobia (54%), followed by nausea (28%), and phonophobia (15%).
In Study 1, the percentage of patients achieving headache pain freedom and MBS freedom two hours after a single dose was statistically significantly greater in patients who received NURTEC ODT compared to those who received placebo (Table 1).
| Study 1 | ||
|---|---|---|
| NURTEC ODT 75 mg | Placebo | |
| ||
Pain Free at 2 hours | ||
n/N* | 142/669 | 74/682 |
% Responders | 21.2 | 10.9 |
Difference from placebo (%) | 10.3 | |
p-value | <0.001 | |
MBS Free at 2 hours | ||
n/N* | 235/669 | 183/682 |
% Responders | 35.1 | 26.8 |
Difference from placebo (%) | 8.3 | |
p-value | 0.001 | |
Figure 1 presents the percentage of patients achieving migraine pain freedom within 2 hours following treatment in Study 1.
Figure 1: Percentage of Patients Achieving Pain Freedom within 2 Hours in Study 1
Figure 2 presents the percentage of patients achieving MBS freedom within 2 hours in Study 1.
Figure 2: Percentage of Patients Achieving MBS Freedom within 2 Hours in Study 1
In Study 1, statistically significant effects of NURTEC ODT compared to placebo were demonstrated for the additional efficacy endpoints of pain relief at 2 hours, sustained pain freedom 2-48 hours, use of rescue medication within 24 hours, and the percentage of patients reporting normal function at two hours after dosing (Table 2). Pain relief was defined as a reduction in migraine pain from moderate or severe severity to mild or none. The measurement of the percentage of patients reporting normal function at two hours after dosing was derived from a single item questionnaire, asking patients to select one response on a 4-point scale; normal function, mild impairment, severe impairment, or required bedrest.
| Study 1 | ||
|---|---|---|
| NURTEC ODT 75 mg | Placebo | |
Pain Relief at 2 hours | ||
n/N* | 397/669 | 295/682 |
% Responders | 59.3 | 43.3 |
Difference from placebo | 16.1 | |
p-value | <0.001 | |
Sustained Pain Freedom 2-48 hours | ||
n/N* | 90/669 | 37/682 |
% Responders | 13.5 | 5.4 |
Difference from placebo | 8.0 | |
p-value | <0.001 | |
Use of Rescue Medication within 24 hours† | ||
n/N* | 95/669 | 199/682 |
% Responders | 14.2 | 29.2 |
Difference from placebo | -15.0 | |
p-value | <0.001 | |
Percentage of Patients Reporting Normal Function at 2 hours | ||
n/N* | 255/669 | 176/682 |
% Responders | 38.1 | 25.8 |
Difference from placebo | 12.3 | |
p-value | <0.001 | |
The incidence of photophobia and phonophobia was reduced following administration of NURTEC ODT 75 mg as compared to placebo.
14.2 Preventive Treatment of Episodic Migraine
The efficacy of NURTEC ODT for the preventive treatment of episodic migraine in adults was demonstrated in one randomized, double-blind, placebo-controlled trial of a different oral dosage form of rimegepant (Study 2; NCT03732638).
Study 2 enrolled adult patients with at least a 1-year history of migraine (with or without aura). Patients experienced an average of 10.9 headache days during the 28-day observational period, which included an average of 10.2 migraine days, prior to randomization into the trial. Patients were randomized to receive every other day dosing of rimegepant 75 mg (N=373) or placebo (N=374) for 12 weeks. Patients were allowed to use acute headache treatments (i.e., triptans, NSAIDs, acetaminophen, antiemetics, muscle relaxants, and aspirin) as needed. Approximately 10% of patients were taking one preventive medication for migraine at baseline. The use of a concomitant medication that acts on the CGRP pathway was not permitted for either the acute or preventive treatment of migraine.
The study excluded patients with myocardial infarction, acute coronary syndrome, percutaneous coronary intervention, cardiac surgery, stroke, or transient ischemic attack within six months of screening.
The primary efficacy endpoint for Study 2 was the change from baseline in the mean number of monthly migraine days (MMDs) during Weeks 9 through 12 of the double-blind treatment phase.
The percentage of patients who achieved at least a 50% reduction from baseline in moderate to severe MMDs during Weeks 9 through 12 of the double-blind treatment phase compared to placebo was also evaluated. Rimegepant 75 mg dosed every other day demonstrated statistically significant improvements for these efficacy endpoints compared to placebo, as summarized in Table 3.
| Rimegepant 75 mg Every Other Day | Placebo Every Other Day | |
|---|---|---|
Monthly Migraine Days (MMD), Weeks 9-12 | N=348 | N=347 |
Change from baseline | -4.3 | -3.5 |
Change from placebo | -0.8 | |
p-value | 0.010 | |
≥ 50% Responders (Moderate to Severe MMDs), Weeks 9-12 | N=348 | N=347 |
% Responders | 49.1 | 41.5 |
Difference from placebo | 7.6 | |
p-value | 0.044 |
Figure 3: Change from Baseline in Monthly Migraine Days in Study 2a
aLeast-square means and 95% confidence intervals are presented.
Figure 4: Distribution of Change from Baseline in Mean Monthly Migraine Days at Month 3 by Treatment Group in Study 2a
aFigure excludes patients with missing data.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.