gemcitabine injection powder 1GM, 200MG Clinical Studies
14 CLINICAL STUDIES
14.1 Ovarian Cancer
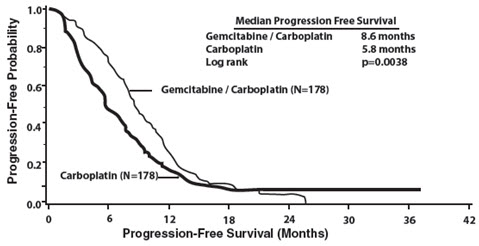
The efficacy of gemcitabine was evaluated in a randomized trial (Study 1) conducted in women with advanced ovarian cancer that had relapsed at least 6 months after first-line platinum-based therapy. Patients were randomized to receive either gemcitabine 1,000 mg/m2 on Days 1 and 8 of each 21-day cycle with carboplatin AUC 4 on Day 1 after gemcitabine administration (n=178) or carboplatin AUC 5 on Day 1 of each 21-day cycle (n=178). The major efficacy outcome measure was progression-free survival (PFS).
A total of 356 patients were enrolled. Demographics and baseline characteristics are shown in Table 16.
Efficacy results are presented in Table 17 and Figure 1. The addition of gemcitabine to carboplatin resulted in statistically significant improvements in PFS and overall response rate. Approximately 75% of patients in each arm received additional chemotherapy for disease progression; 13 of 120 patients in the carboplatin alone arm received gemcitabine for treatment of disease progression. There was no significant difference in overall survival between the treatment arms.
| Gemcitabine/Carboplatin (N=178) | Carboplatin (N=178) | |
|---|---|---|
| Median age, years | 59 | 58 |
| Range | 36 to 78 | 21 to 81 |
| Baseline ECOG performance status 0–1* | 94% | 95% |
| Disease Status | ||
| Evaluable | 8% | 3% |
| Bidimensionally measurable | 92% | 96% |
| Platinum-free interval† | ||
| 6–12 months | 40% | 40% |
| >12 months | 59% | 60% |
| First-line therapy | ||
| Platinum-taxane combination | 70% | 71% |
| Platinum-non-taxane combination | 29% | 28% |
| Platinum monotherapy | 1% | 1% |
| Efficacy Parameter | Gemcitabine/Carboplatin (N=178) | Carboplatin (N=178) |
|---|---|---|
| ||
| Progression-Free Survival | ||
| Median (95% CI*) in months | 8.6 (8.0, 9.7) | 5.8 (5.2, 7.1) |
| Hazard Ratio (95% CI) | 0.72 (0.57, 0.90) | |
| p-value† | p=0.0038 | |
| Overall Survival | ||
| Median (95% CI) in months | 18.0 (16.2, 20.3) | 17.3 (15.2, 19.3) |
| Hazard Ratio (95% CI) | 0.98 (0.78, 1.24) | |
| p-value† | p=0.8977 | |
| Overall Response Rate by Investigator Review | 47.2% | 30.9% |
| p-value‡ | p=0.0016 | |
| CR§ | 14.6% | 6.2% |
| PR with PRNM¶ | 32.6% | 24.7% |
| Overall Response Rate# by Independent Review | 46.3% | 35.6% |
| p-value‡ | p=0.11 | |
| CR§ | 9.1% | 4.0% |
| PR with PRNM¶ | 37.2% | 31.7% |
| Figure 1: Kaplan-Meier Curves for Progression-Free Survival in Study 1 |
|---|
 |
14.2 Breast Cancer
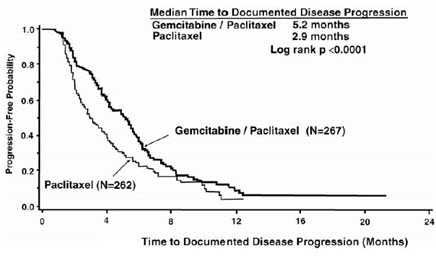
The efficacy of gemcitabine was evaluated in a multinational, randomized, open-label trial (Study 2) conducted in women receiving initial treatment for metastatic breast cancer and who have received prior adjuvant/neoadjuvant anthracycline chemotherapy unless clinically contraindicated. Patients were randomized to receive either gemcitabine 1,250 mg/m2 on Days 1 and 8 of each 21-day cycle with paclitaxel 175 mg/m2 administered on Day 1 before gemcitabine administration (n=267) or paclitaxel 175 mg/m2 on Day 1 of each 21-day cycle (n=262). The major efficacy outcome measure was time to documented disease progression.
A total of 529 patients were enrolled. Demographic and baseline characteristics were similar between treatment arms (Table 18).
Efficacy results are presented in Table 19 and Figure 2. The addition of gemcitabine to paclitaxel resulted in statistically significant improvement in time to documented disease progression and overall response rate compared to paclitaxel alone. There was no significant difference in overall survival.
| Gemcitabine/Paclitaxel (N=267) | Paclitaxel (N=262) | |
|---|---|---|
| ||
| Median age (years) | 53 | 52 |
| Range | 26 to 83 | 26 to 75 |
| Metastatic disease | 97% | 97% |
| Baseline KPS* ≥90 | 70% | 74% |
| Number of tumor sites | ||
| 1–2 | 57% | 59% |
| ≥3 | 43% | 41% |
| Visceral disease | 73% | 73% |
| Prior anthracycline | 97% | 96% |
| Efficacy Parameter | Gemcitabine/Paclitaxel (N=267) | Paclitaxel (N=262) |
|---|---|---|
| Time to Documented Disease Progression* | ||
| Median (95% CI) in months | 5.2 (4.2, 5.6) | 2.9 (2.6, 3.7) |
| Hazard Ratio (95% CI) | 0.650 (0.524, 0.805) | |
| p-value | p<0.0001 | |
| Overall Survival† | ||
| Median (95% CI) in months | 18.6 (16.5, 20.7) | 15.8 (14.1, 17.3) |
| Hazard Ratio (95% CI) | 0.86 (0.71, 1.04) | |
| p-value | Not Significant | |
| Overall Response Rate | 40.8% | 22.1% |
| (95% CI) | (34.9, 46.7) | (17.1, 27.2) |
| p-value | p<0.0001 | |
| Figure 2: Kaplan-Meier Curves for Time to Documented Disease Progression in Study 2 |
|---|
 |
14.3 Non-Small Cell Lung Cancer
The efficacy of gemcitabine was evaluated in two randomized, multicenter trials.
Study 3: 28-Day Schedule
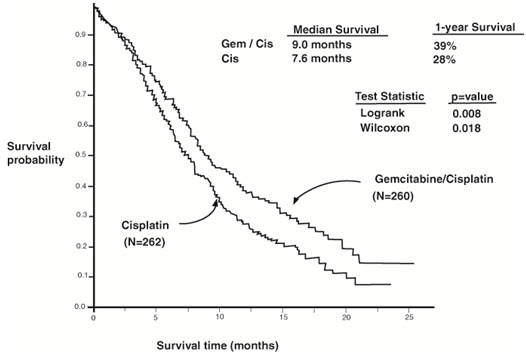
A multinational, randomized trial (Study 3) compared gemcitabine with cisplatin to cisplatin alone in the treatment of patients with inoperable Stage IIIA, IIIB, or IV NSCLC who had not received prior chemotherapy. Patients were randomized to receive either gemcitabine 1,000 mg/m2 on Days 1, 8, and 15 of each 28-day cycle with cisplatin 100 mg/m2 on Day 1 after gemcitabine administration (N=260) or cisplatin 100 mg/m2 on Day 1 of each 28-day cycle (N=262). The major efficacy outcome measure was overall survival.
A total of 522 patients were enrolled. Demographics and baseline characteristics (Table 20) were similar between arms with the exception of histologic subtype of NSCLC, with 48% of patients on the cisplatin arm and 37% of patients on the gemcitabine with cisplatin arm having adenocarcinoma.
Efficacy results are presented in Table 21 and Figure 3.
Study 4: 21-Day Schedule
A randomized (1:1), multicenter trial (Study 4) was conducted in patients with Stage IIIB or IV NSCLC. Patients were randomized to receive either gemcitabine 1,250 mg/m2 on Days 1 and 8 of each 21-day cycle with cisplatin 100 mg/m2 on Day 1 after gemcitabine administration or etoposide 100 mg/m2 intravenously on Days 1, 2, and 3 with cisplatin 100 mg/m2 on Day 1 of each 21-day cycle. The major efficacy outcome measure was response rate.
A total of 135 patients were enrolled. Demographics and baseline characteristics are summarized in Table 20.
Efficacy results are presented in Table 21. There was no significant difference in survival between the two treatment arms. The median survival was 8.7 months for the gemcitabine with cisplatin arm versus 7 months for the etoposide with cisplatin arm. Median time to disease progression for the gemcitabine with cisplatin arm was 5 months compared to 4.1 months on the etoposide with cisplatin arm (Log rank p=0.015, two-sided). The objective response rate for the gemcitabine with cisplatin arm was 33% compared to 14% on the etoposide with cisplatin arm (Fisher's Exact p=0.01, two-sided).
| Trial | 28-day Schedule (Study 3) | 21-day Schedule (Study 4) | ||
|---|---|---|---|---|
| Gemcitabine/Cisplatin (N=260) | Cisplatin (N=262) | Gemcitabine/Cisplatin (N=69) | Etoposide/Cisplatin (N=66) | |
| Male | 70% | 71% | 93% | 92% |
| Median age, years | 62 | 63 | 58 | 60 |
| Range | 36 to 88 | 35 to 79 | 33 to 76 | 35 to 75 |
| Stage IIIA | 7% | 7% | N/A* | N/A* |
| Stage IIIB | 26% | 23% | 48% | 52% |
| Stage IV | 67% | 70% | 52% | 49% |
| Baseline KPS† 70 to 80 | 41% | 44% | 45% | 52% |
| Baseline KPS† 90 to 100 | 57% | 55% | 55% | 49% |
| Trial | 28-day Schedule (Study 3) | 21-day Schedule (Study 4) | ||
|---|---|---|---|---|
| Efficacy Parameter | Gemcitabine/Cisplatin (N=260) | Cisplatin (N=262) | Gemcitabine/Cisplatin (N=69) | Etoposide/Cisplatin (N=66) |
| Survival | ||||
| Median (95% CI*) in months | 9.0 (8.2, 11.0) | 7.6 (6.6, 8.8) | 8.7 (7.8, 10.1) | 7.0 (6.0, 9.7) |
| p-valuef | p=0.008 | p=0.18 | ||
| Time to Disease Progression | ||||
| Median (95% CI*) in months | 5.2 (4.2, 5.7) | 3.7 (3.0, 4.3) | 5.0 (4.2, 6.4) | 4.1 (2.4, 4.5) |
| p-value† | p=0.009 | p=0.015 | ||
| Tumor Response | 26% | 10% | 33% | 14% |
| p-value† | p<0.0001 | p=0.01 | ||
| Figure 3: Kaplan-Meier Curves for Overall Survival in Study 3 |
|---|
 |
14.4 Pancreatic Cancer
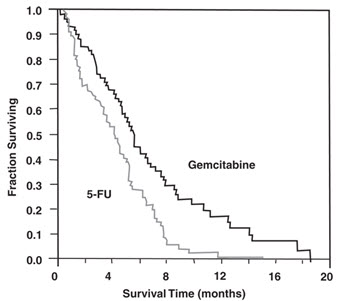
The efficacy of gemcitabine was evaluated in two trials (Studies 5 and 6), a randomized, single-blind, two-arm, active-controlled trial (Study 5) conducted in patients with locally advanced or metastatic pancreatic cancer who had received no prior chemotherapy and in a single-arm, open-label, multicenter trial (Study 6) conducted in patients with locally advanced or metastatic pancreatic cancer previously treated with fluorouracil or a fluorouracil-containing regimen. In Study 5, patients were randomized to receive either gemcitabine 1,000 mg/m2 intravenously over 30 minutes once weekly for 7 weeks followed by a one-week rest, then once weekly for 3 consecutive weeks every 28-days in subsequent cycles (n=63) or fluorouracil 600 mg/m2 intravenously over 30 minutes once weekly (n=63). In Study 6, all patients received gemcitabine 1,000 mg/m2 intravenously over 30 minutes once weekly for 7 weeks followed by a one-week rest, then once weekly for 3 consecutive weeks every 28-days in subsequent cycles.
The major efficacy outcome measure in both trials was "clinical benefit response". A patient was considered to have had a clinical benefit response if either of the following occurred:
- The patient achieved a ≥50% reduction in pain intensity (Memorial Pain Assessment Card) or analgesic consumption, or a 20-point or greater improvement in performance status (Karnofsky Performance Status) for a period of at least 4 consecutive weeks, without showing any sustained worsening in any of the other parameters. Sustained worsening was defined as 4 consecutive weeks with either any increase in pain intensity or analgesic consumption or a 20-point decrease in performance status occurring during the first 12 weeks of therapy.
OR - The patient was stable on all of the aforementioned parameters and showed a marked, sustained weight gain (≥7% increase maintained for ≥4 weeks) not due to fluid accumulation.
Study 5 enrolled 126 patients. Demographics and baseline characteristics were similar between the arms (Table 22).
The efficacy results are shown in Table 23 and Figure 4. Patients treated with gemcitabine had statistically significant increases in clinical benefit response, survival, and time to disease progression compared to those randomized to receive fluorouracil. No confirmed objective tumor responses were observed in either treatment arm.
| Gemcitabine (N=63) | Fluorouracil (N=63) | |
|---|---|---|
| ||
| Male | 54% | 54% |
| Median age, years | 62 | 61 |
| Range | 37 to 79 | 36 to 77 |
| Stage IV disease | 71% | 76% |
| Baseline KPS* ≤70 | 70% | 68% |
| Efficacy Parameter | Gemcitabine (N=63) | Fluorouracil (N=63) |
|---|---|---|
| ||
| Clinical Benefit Response p-value* | 22.2% | 4.8% |
| p=0.004 | ||
| Overall Survival | ||
| Median (95% CI) in months | 5.7 (4.7, 6.9) | 4.2 (3.1, 5.1) |
| p-value* | p=0.0009 | |
| Time to Disease Progression | ||
| Median (95% CI) in months | 2.1 (1.9, 3.4) | 0.9 (0.9, 1.1) |
| p-value* | p=0.0013 | |
| Figure 4: Kaplan-Meier Curves for Overall Survival in Study 5 |
|---|
 |
Find gemcitabine injection powder 1GM, 200MG medical information:
Find gemcitabine injection powder 1GM, 200MG medical information:
gemcitabine injection powder 1GM, 200MG Quick Finder
Health Professional Information
Clinical Studies
14 CLINICAL STUDIES
14.1 Ovarian Cancer
The efficacy of gemcitabine was evaluated in a randomized trial (Study 1) conducted in women with advanced ovarian cancer that had relapsed at least 6 months after first-line platinum-based therapy. Patients were randomized to receive either gemcitabine 1,000 mg/m2 on Days 1 and 8 of each 21-day cycle with carboplatin AUC 4 on Day 1 after gemcitabine administration (n=178) or carboplatin AUC 5 on Day 1 of each 21-day cycle (n=178). The major efficacy outcome measure was progression-free survival (PFS).
A total of 356 patients were enrolled. Demographics and baseline characteristics are shown in Table 16.
Efficacy results are presented in Table 17 and Figure 1. The addition of gemcitabine to carboplatin resulted in statistically significant improvements in PFS and overall response rate. Approximately 75% of patients in each arm received additional chemotherapy for disease progression; 13 of 120 patients in the carboplatin alone arm received gemcitabine for treatment of disease progression. There was no significant difference in overall survival between the treatment arms.
| Gemcitabine/Carboplatin (N=178) | Carboplatin (N=178) | |
|---|---|---|
| Median age, years | 59 | 58 |
| Range | 36 to 78 | 21 to 81 |
| Baseline ECOG performance status 0–1* | 94% | 95% |
| Disease Status | ||
| Evaluable | 8% | 3% |
| Bidimensionally measurable | 92% | 96% |
| Platinum-free interval† | ||
| 6–12 months | 40% | 40% |
| >12 months | 59% | 60% |
| First-line therapy | ||
| Platinum-taxane combination | 70% | 71% |
| Platinum-non-taxane combination | 29% | 28% |
| Platinum monotherapy | 1% | 1% |
| Efficacy Parameter | Gemcitabine/Carboplatin (N=178) | Carboplatin (N=178) |
|---|---|---|
| ||
| Progression-Free Survival | ||
| Median (95% CI*) in months | 8.6 (8.0, 9.7) | 5.8 (5.2, 7.1) |
| Hazard Ratio (95% CI) | 0.72 (0.57, 0.90) | |
| p-value† | p=0.0038 | |
| Overall Survival | ||
| Median (95% CI) in months | 18.0 (16.2, 20.3) | 17.3 (15.2, 19.3) |
| Hazard Ratio (95% CI) | 0.98 (0.78, 1.24) | |
| p-value† | p=0.8977 | |
| Overall Response Rate by Investigator Review | 47.2% | 30.9% |
| p-value‡ | p=0.0016 | |
| CR§ | 14.6% | 6.2% |
| PR with PRNM¶ | 32.6% | 24.7% |
| Overall Response Rate# by Independent Review | 46.3% | 35.6% |
| p-value‡ | p=0.11 | |
| CR§ | 9.1% | 4.0% |
| PR with PRNM¶ | 37.2% | 31.7% |
| Figure 1: Kaplan-Meier Curves for Progression-Free Survival in Study 1 |
|---|
 |
14.2 Breast Cancer
The efficacy of gemcitabine was evaluated in a multinational, randomized, open-label trial (Study 2) conducted in women receiving initial treatment for metastatic breast cancer and who have received prior adjuvant/neoadjuvant anthracycline chemotherapy unless clinically contraindicated. Patients were randomized to receive either gemcitabine 1,250 mg/m2 on Days 1 and 8 of each 21-day cycle with paclitaxel 175 mg/m2 administered on Day 1 before gemcitabine administration (n=267) or paclitaxel 175 mg/m2 on Day 1 of each 21-day cycle (n=262). The major efficacy outcome measure was time to documented disease progression.
A total of 529 patients were enrolled. Demographic and baseline characteristics were similar between treatment arms (Table 18).
Efficacy results are presented in Table 19 and Figure 2. The addition of gemcitabine to paclitaxel resulted in statistically significant improvement in time to documented disease progression and overall response rate compared to paclitaxel alone. There was no significant difference in overall survival.
| Gemcitabine/Paclitaxel (N=267) | Paclitaxel (N=262) | |
|---|---|---|
| ||
| Median age (years) | 53 | 52 |
| Range | 26 to 83 | 26 to 75 |
| Metastatic disease | 97% | 97% |
| Baseline KPS* ≥90 | 70% | 74% |
| Number of tumor sites | ||
| 1–2 | 57% | 59% |
| ≥3 | 43% | 41% |
| Visceral disease | 73% | 73% |
| Prior anthracycline | 97% | 96% |
| Efficacy Parameter | Gemcitabine/Paclitaxel (N=267) | Paclitaxel (N=262) |
|---|---|---|
| Time to Documented Disease Progression* | ||
| Median (95% CI) in months | 5.2 (4.2, 5.6) | 2.9 (2.6, 3.7) |
| Hazard Ratio (95% CI) | 0.650 (0.524, 0.805) | |
| p-value | p<0.0001 | |
| Overall Survival† | ||
| Median (95% CI) in months | 18.6 (16.5, 20.7) | 15.8 (14.1, 17.3) |
| Hazard Ratio (95% CI) | 0.86 (0.71, 1.04) | |
| p-value | Not Significant | |
| Overall Response Rate | 40.8% | 22.1% |
| (95% CI) | (34.9, 46.7) | (17.1, 27.2) |
| p-value | p<0.0001 | |
| Figure 2: Kaplan-Meier Curves for Time to Documented Disease Progression in Study 2 |
|---|
 |
14.3 Non-Small Cell Lung Cancer
The efficacy of gemcitabine was evaluated in two randomized, multicenter trials.
Study 3: 28-Day Schedule
A multinational, randomized trial (Study 3) compared gemcitabine with cisplatin to cisplatin alone in the treatment of patients with inoperable Stage IIIA, IIIB, or IV NSCLC who had not received prior chemotherapy. Patients were randomized to receive either gemcitabine 1,000 mg/m2 on Days 1, 8, and 15 of each 28-day cycle with cisplatin 100 mg/m2 on Day 1 after gemcitabine administration (N=260) or cisplatin 100 mg/m2 on Day 1 of each 28-day cycle (N=262). The major efficacy outcome measure was overall survival.
A total of 522 patients were enrolled. Demographics and baseline characteristics (Table 20) were similar between arms with the exception of histologic subtype of NSCLC, with 48% of patients on the cisplatin arm and 37% of patients on the gemcitabine with cisplatin arm having adenocarcinoma.
Efficacy results are presented in Table 21 and Figure 3.
Study 4: 21-Day Schedule
A randomized (1:1), multicenter trial (Study 4) was conducted in patients with Stage IIIB or IV NSCLC. Patients were randomized to receive either gemcitabine 1,250 mg/m2 on Days 1 and 8 of each 21-day cycle with cisplatin 100 mg/m2 on Day 1 after gemcitabine administration or etoposide 100 mg/m2 intravenously on Days 1, 2, and 3 with cisplatin 100 mg/m2 on Day 1 of each 21-day cycle. The major efficacy outcome measure was response rate.
A total of 135 patients were enrolled. Demographics and baseline characteristics are summarized in Table 20.
Efficacy results are presented in Table 21. There was no significant difference in survival between the two treatment arms. The median survival was 8.7 months for the gemcitabine with cisplatin arm versus 7 months for the etoposide with cisplatin arm. Median time to disease progression for the gemcitabine with cisplatin arm was 5 months compared to 4.1 months on the etoposide with cisplatin arm (Log rank p=0.015, two-sided). The objective response rate for the gemcitabine with cisplatin arm was 33% compared to 14% on the etoposide with cisplatin arm (Fisher's Exact p=0.01, two-sided).
| Trial | 28-day Schedule (Study 3) | 21-day Schedule (Study 4) | ||
|---|---|---|---|---|
| Gemcitabine/Cisplatin (N=260) | Cisplatin (N=262) | Gemcitabine/Cisplatin (N=69) | Etoposide/Cisplatin (N=66) | |
| Male | 70% | 71% | 93% | 92% |
| Median age, years | 62 | 63 | 58 | 60 |
| Range | 36 to 88 | 35 to 79 | 33 to 76 | 35 to 75 |
| Stage IIIA | 7% | 7% | N/A* | N/A* |
| Stage IIIB | 26% | 23% | 48% | 52% |
| Stage IV | 67% | 70% | 52% | 49% |
| Baseline KPS† 70 to 80 | 41% | 44% | 45% | 52% |
| Baseline KPS† 90 to 100 | 57% | 55% | 55% | 49% |
| Trial | 28-day Schedule (Study 3) | 21-day Schedule (Study 4) | ||
|---|---|---|---|---|
| Efficacy Parameter | Gemcitabine/Cisplatin (N=260) | Cisplatin (N=262) | Gemcitabine/Cisplatin (N=69) | Etoposide/Cisplatin (N=66) |
| Survival | ||||
| Median (95% CI*) in months | 9.0 (8.2, 11.0) | 7.6 (6.6, 8.8) | 8.7 (7.8, 10.1) | 7.0 (6.0, 9.7) |
| p-valuef | p=0.008 | p=0.18 | ||
| Time to Disease Progression | ||||
| Median (95% CI*) in months | 5.2 (4.2, 5.7) | 3.7 (3.0, 4.3) | 5.0 (4.2, 6.4) | 4.1 (2.4, 4.5) |
| p-value† | p=0.009 | p=0.015 | ||
| Tumor Response | 26% | 10% | 33% | 14% |
| p-value† | p<0.0001 | p=0.01 | ||
| Figure 3: Kaplan-Meier Curves for Overall Survival in Study 3 |
|---|
 |
14.4 Pancreatic Cancer
The efficacy of gemcitabine was evaluated in two trials (Studies 5 and 6), a randomized, single-blind, two-arm, active-controlled trial (Study 5) conducted in patients with locally advanced or metastatic pancreatic cancer who had received no prior chemotherapy and in a single-arm, open-label, multicenter trial (Study 6) conducted in patients with locally advanced or metastatic pancreatic cancer previously treated with fluorouracil or a fluorouracil-containing regimen. In Study 5, patients were randomized to receive either gemcitabine 1,000 mg/m2 intravenously over 30 minutes once weekly for 7 weeks followed by a one-week rest, then once weekly for 3 consecutive weeks every 28-days in subsequent cycles (n=63) or fluorouracil 600 mg/m2 intravenously over 30 minutes once weekly (n=63). In Study 6, all patients received gemcitabine 1,000 mg/m2 intravenously over 30 minutes once weekly for 7 weeks followed by a one-week rest, then once weekly for 3 consecutive weeks every 28-days in subsequent cycles.
The major efficacy outcome measure in both trials was "clinical benefit response". A patient was considered to have had a clinical benefit response if either of the following occurred:
- The patient achieved a ≥50% reduction in pain intensity (Memorial Pain Assessment Card) or analgesic consumption, or a 20-point or greater improvement in performance status (Karnofsky Performance Status) for a period of at least 4 consecutive weeks, without showing any sustained worsening in any of the other parameters. Sustained worsening was defined as 4 consecutive weeks with either any increase in pain intensity or analgesic consumption or a 20-point decrease in performance status occurring during the first 12 weeks of therapy.
OR - The patient was stable on all of the aforementioned parameters and showed a marked, sustained weight gain (≥7% increase maintained for ≥4 weeks) not due to fluid accumulation.
Study 5 enrolled 126 patients. Demographics and baseline characteristics were similar between the arms (Table 22).
The efficacy results are shown in Table 23 and Figure 4. Patients treated with gemcitabine had statistically significant increases in clinical benefit response, survival, and time to disease progression compared to those randomized to receive fluorouracil. No confirmed objective tumor responses were observed in either treatment arm.
| Gemcitabine (N=63) | Fluorouracil (N=63) | |
|---|---|---|
| ||
| Male | 54% | 54% |
| Median age, years | 62 | 61 |
| Range | 37 to 79 | 36 to 77 |
| Stage IV disease | 71% | 76% |
| Baseline KPS* ≤70 | 70% | 68% |
| Efficacy Parameter | Gemcitabine (N=63) | Fluorouracil (N=63) |
|---|---|---|
| ||
| Clinical Benefit Response p-value* | 22.2% | 4.8% |
| p=0.004 | ||
| Overall Survival | ||
| Median (95% CI) in months | 5.7 (4.7, 6.9) | 4.2 (3.1, 5.1) |
| p-value* | p=0.0009 | |
| Time to Disease Progression | ||
| Median (95% CI) in months | 2.1 (1.9, 3.4) | 0.9 (0.9, 1.1) |
| p-value* | p=0.0013 | |
| Figure 4: Kaplan-Meier Curves for Overall Survival in Study 5 |
|---|
 |
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.