Sodium Chloride 0.9% ADD-Vantage Diluent
()
Find Sodium Chloride 0.9% ADD-Vantage Diluent medical information:
Find Sodium Chloride 0.9% ADD-Vantage Diluent medical information:
Sodium Chloride 0.9% ADD-Vantage Diluent Quick Finder
Indications and Usage
INDICATIONS AND USAGE
Intravenous solutions containing sodium chloride are indicated for parenteral replenishment of fluid and sodium chloride as required by the clinical condition of the patient.
In this dosage form, Sodium Chloride Injection, USP is intended to be used as a diluent for the contents of an ADD-Vantage vial, or single-dose vials with 20 mm closure using the ADD-Vantage ADDAPTOR™.
Dosage and Administration
DOSAGE AND ADMINISTRATION
The dose is dependent upon the age, weight and clinical condition of the patient.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS.
Contraindications
Warnings and Precautions
WARNINGS
Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency and in clinical states in which there exists edema with sodium retention.
Excessive administration of potassium-free solutions may result in significant hypokalemia.
In patients with diminished renal function, administration of solutions containing sodium ions may result in sodium retention.
The intravenous administration of these solutions can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
The risk of dilutional states is inversely proportional to the electrolyte concentrations of administered parenteral solutions. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of such solutions.
PRECAUTIONS
General
Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Use of a vented intravenous administration set with the vent in the open position could result in air embolism. Vented intravenous administration sets with the vent in the open position should not be used with flexible plastic containers.
Laboratory Tests
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Adverse Reactions
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
Drug Interactions
DRUG INTERACTIONS
Caution must be exercised in the administration of parenteral fluids, especially those containing sodium ions to patients receiving corticosteroids or corticotropin.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Studies with Sodium Chloride Injection, USP have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Pregnancy: Teratogenic effects
Animal reproduction studies have not been conducted with Sodium Chloride Injection, USP. It is not known whether sodium chloride injection can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Sodium Chloride Injection, USP should be given to a pregnant woman only if clearly needed.
Labor and Delivery: Studies have not been conducted to evaluate the effects of Sodium Chloride Injection, USP on labor and delivery. Caution should be exercised when administering this drug during labor and delivery.
Nursing Mothers: It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Sodium Chloride Injection, USP is administered to a nursing mother.
Pediatric Use: Safety and effectiveness in children have not been established.
Geriatric Use: Clinical studies of Sodium Chloride Injection, USP did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Do not administer unless solution is clear and container is undamaged. Discard unused portion.
Overdosage
OVERDOSAGE
In the event of overhydration or solute overload, re-evaluate the patient and institute appropriate corrective measures. See WARNINGS, PRECAUTIONS, and ADVERSE REACTIONS.
Description
DESCRIPTION
Sodium Chloride Injection, USP solutions are sterile and nonpyrogenic. They are parenteral solutions containing various concentrations of sodium chloride in water for injection intended for intravenous administration after admixing with an ADD-Vantage vial, or single-dose powdered drug vials with 20 mm closure using the ADD-Vantage ADDAPTOR™ (WARNING: DO NOT USE WITH CHEMOTHERAPY AGENTS).
Each 100 mL of 0.45% Sodium Chloride Injection, USP contains 450 mg sodium chloride in water for injection. Electrolytes per 1000 mL: sodium 77 mEq; chloride 77 mEq. The osmolarity is 154 mOsmol/L (calc.), which is hypotonic.
Each 100 mL of 0.9% Sodium Chloride Injection, USP contains 900 mg sodium chloride in water for injection. Electrolytes per 1000 mL: sodium 154 mEq; chloride 154 mEq. The osmolarity is 308 mOsmol/L (calc.), which is isotonic.
The pH for both concentrations is 5.6 (4.5 to 7.0).
The solutions contain no bacteriostat, antimicrobial agent or added buffer and each is intended only as a single-dose injection. When smaller doses are required the unused portion should be discarded.
The solutions are parenteral fluid and electrolyte replenishers.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials. Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
Clinical Pharmacology
CLINICAL PHARMACOLOGY
When administered intravenously, these solutions provide a source of water and electrolytes.
Solutions which provide an isotonic concentration of sodium chloride are suitable for parenteral maintenance or replacement of water and electrolyte requirements.
Isotonic concentrations of sodium chloride are suitable for parenteral replacement of chloride losses that exceed or equal the sodium loss. Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl‾) ions. Sodium (Na+) is the principal cation of the extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl‾) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in the red blood cells. The distribution and excretion of sodium (Na+) and chloride (Cl‾) are largely under the control of the kidney which maintains a balance between intake and output.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirements range from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
How Supplied/Storage and Handling
HOW SUPPLIED
Unit of Sale | Concentration | Each |
NDC 0409-7132-66 Case of 50 | 0.45% | NDC 0409-7132-68 50 mL bags |
NDC 0409-7132-67 Case of 50 | 0.45% | NDC 0409-7132-69 100 mL bags |
NDC 0409-7132-02 Case of 24 | 0.45% | NDC 0409-7132-04 250 mL bags |
NDC 0409-7101-66 Case of 50 | 0.9% | NDC 0409-7101-68 50 mL bags |
NDC 0409-7101-67 Case of 50 | 0.9% | NDC 0409-7101-69 100 mL bags |
NDC 0409-7101-02 Case of 24 | 0.9% | NDC 0409-7101-04 250 mL bags |
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
INSTRUCTIONS FOR USE WITH ADD-VANTAGE VIAL
These instructions for use should be made available to the individuals who perform the reconstitution steps.
To Open:
Peel overwrap at corner and remove solution container. Use unit within 30 days of opening overwrap, as long as the use date does not exceed the printed expiration date. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Assemble Vial and Flexible Diluent Container:
(Use Aseptic Technique)
- 1.
- Remove the protective covers from the top of the vial and the vial port on the diluent container as follows:
- a.
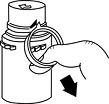
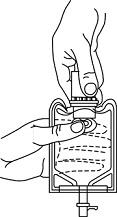
- To remove the breakaway vial cap, swing the pull ring over the top of the vial and pull down far enough to start the opening (SEE FIGURE 1), then pull straight up to remove the cap. (SEE FIGURE 2.)
NOTE: Once the breakaway cap has been removed, do not access vial with syringe.
Fig. 1 Fig. 2
- b.
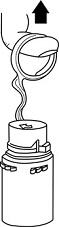
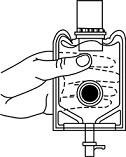
- To remove the vial port cover, grasp the tab on the pull ring, pull up to break the three tie strings, then pull back to remove the cover. (SEE FIGURE 3.)
- 2.
- Screw the vial into the vial port until it will go no further. THE VIAL MUST BE SCREWED IN TIGHTLY TO ASSURE A SEAL. This occurs approximately 1/2 turn (180°) after the first audible click. (SEE FIGURE 4.) The clicking sound does not assure a seal; the vial must be turned as far as it will go.
NOTE: Once vial is seated, do not attempt to remove. (SEE FIGURE 4.) - 3.
- Recheck the vial to assure that it is tight by trying to turn it further in the direction of assembly.
- 4.
- Label appropriately.
Fig. 3 Fig. 4
To Reconstitute the Drug:
- 1.
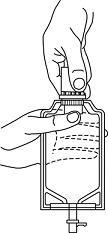
- Squeeze the bottom of the diluent container gently to inflate the portion of the container surrounding the end of the drug vial.
- 2.
- With the other hand, push the drug vial down into the container telescoping the walls of the container. Grasp the inner cap of the vial through the walls of the container. (SEE FIGURE 5.)
- 3.
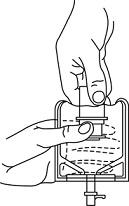
- Pull the inner cap from the drug vial. (SEE FIGURE 6.) Verify that the rubber stopper has been pulled out, allowing the drug and diluent to mix.
- 4.
- Mix container contents thoroughly and use within the specified time.
- 5.
- Look through the bottom of the vial to verify that the stopper has been removed and complete mixing has occurred. (SEE FIGURE 7.)
If the rubber stopper is not removed from the vial and medication is not released on the first attempt, the inner cap may be manipulated back into the rubber stopper without removing the drug vial from the diluent container. Repeat steps 3 through 5.
Fig. 5 Fig. 6 Fig. 7
Preparation for Administration:
(Use Aseptic Technique)
- 1.
- Confirm the activation and admixture of vial contents.
- 2.
- Check for leaks by squeezing container firmly. If leaks are found, discard unit as sterility may be impaired.
- 3.
- Close flow control clamp of administration set.
- 4.
- Remove cover from outlet port at bottom of container.
- 5.
- Insert piercing pin of administration set into port with a twisting motion until the pin is firmly seated. NOTE: See full directions on administration set carton.
- 6.
- Lift the free end of the hanger loop on the bottom of the vial, breaking the two tie strings. Bend the loop outward to lock it in the upright position, then suspend container from hanger.
- 7.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- 8.
- Open flow control clamp and clear air from set. Close clamp.
- 9.
- Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
- 10.
- Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible container in series connections.
INSTRUCTIONS FOR USE WITH ADD-VANTAGE ADDAPTOR™
The instructions for use provided with the ADD-Vantage ADDAPTOR™ should be made available to the individuals who perform the reconstitution steps.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA 
LAB-1103-2.0
Revised: 11/2018
Other
Resources
Didn’t find what you were looking for?
Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5Pm ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this product, click the link below to submit your information: Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer product, please call Pfizer Medical Information at (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800)-332-1088.