2 DOSAGE AND ADMINISTRATION
For intramuscular administration only.
2.1 Preparation
Do not mix Prevnar 20 with other vaccines/products in the same syringe.
Step 1. Resuspend drug product
Hold the pre-filled syringe horizontally between the thumb and the forefinger and shake vigorously until the vaccine is a homogeneous white suspension. Do not use the vaccine if it cannot be re-suspended. | 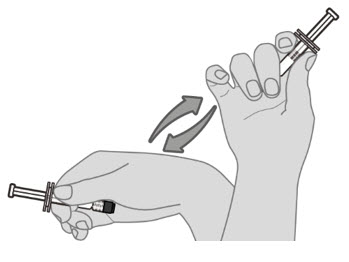 |
Step 2. Visual inspection
Visually inspect the vaccine for large particulate matter and discoloration prior to administration. Do not use if large particulate matter or discoloration is found. If the vaccine is not a homogeneous suspension, repeat Steps 1 and 2. | 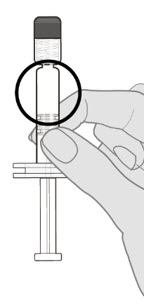 |
Step 3. Remove syringe cap
Remove the syringe cap by slowly turning the cap counterclockwise while holding the Luer lock adapter.
Avoid pressing the syringe plunger rod while removing the syringe cap. | 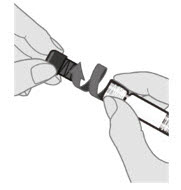 |
Step 4. Attach a sterile needle
Hold the Luer lock adapter and attach a needle appropriate for intramuscular administration to the pre-filled syringe by turning clockwise. |
2.2 Administration
For intramuscular injection only.
Each 0.5-mL dose is to be injected intramuscularly using a sterile needle attached to the supplied pre-filled syringe.
2.3 Vaccination Schedule for Individuals 6 Weeks Through 15 Months of Age
Administer Prevnar 20 as a 4-dose series at 2, 4, 6, and 12 through 15 months of age (and at least 2 months after the third dose). The first dose may be given as early as 6 weeks of age.
2.4 Catch-Up Vaccination Schedule for Unvaccinated Individuals 7 Months Through 17 Years of Age
Individuals 7 months through 17 years of age who have never received a pneumococcal conjugate vaccine may receive Prevnar 20 according to the schedule in Table 1:
Table 1. Catch-Up Vaccination Schedule for Individuals Initiating Vaccination at 7 Months Through 17 Years of Age* |
Age at First Dose | Total Number of 0.5-mL Doses |
7 through 11 months of age | 3† |
12 through 23 months of age | 2‡ |
24 months of age and above | 1 |
2.5 Catch-Up Vaccination Schedule for Individuals Previously Vaccinated With One or More Doses of a Lower Valency Pneumococcal Conjugate Vaccine
Administer a single dose of Prevnar 20 to individuals 15 months through 17 years of age previously vaccinated with one or more doses of a lower valency pneumococcal conjugate vaccine. The dose of Prevnar 20 should be administered at least 8 weeks after the last dose of the lower valency pneumococcal conjugate vaccine.
2.6 Vaccination Schedule for Individuals 18 Years of Age and Older
Prevnar 20 is administered as a single dose.