Levetiracetam Injection Clinical Studies
()
14 CLINICAL STUDIES
All clinical studies supporting the efficacy of levetiracetam utilized oral formulations. The finding of efficacy of levetiracetam injection is based on the results of studies using an oral formulation of levetiracetam, and on the demonstration of comparable bioavailability of the oral and parenteral formulations [see Clinical Pharmacology (12.3)].
14.1 Partial-Onset Seizures
Effectiveness in Partial-Onset Seizures in Adults
The effectiveness of levetiracetam for the treatment of partial-onset seizures in adults was established in three multicenter, randomized, double-blind, placebo-controlled clinical studies in patients who had refractory partial-onset seizures with or without secondary generalization. The tablet formulation was used in all these studies. In these studies, 904 patients were randomized to placebo, 1,000 mg, 2,000 mg, or 3,000 mg/day. Patients enrolled in Study 1 or Study 2 had refractory partial-onset seizures for at least two years and had taken two or more classical AEDs. Patients enrolled in Study 3 had refractory partial-onset seizures for at least 1 year and had taken one classical AED. At the time of the study, patients were taking a stable dose regimen of at least one and could take a maximum of two AEDs. During the baseline period, patients had to have experienced at least two partial-onset seizures during each 4-week period.
Study 1
Study 1 was a double-blind, placebo-controlled, parallel-group study conducted at 41 sites in the United States comparing levetiracetam 1,000 mg/day (N=97), levetiracetam 3,000 mg/day (N=101), and placebo (N=95) given in equally divided doses twice daily. After a prospective baseline period of 12 weeks, patients were randomized to one of the three treatment groups described above. The 18-week treatment period consisted of a 6-week titration period, followed by a 12-week fixed dose evaluation period, during which concomitant AED regimens were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). The results of the analysis of Study 1 are displayed in Table 10.
| Placebo (N=95) | Levetiracetam 1,000 mg/day (N=97) | Levetiracetam 3,000 mg/day (N=101) | |
|---|---|---|---|
| |||
Percent reduction in partial seizure frequency over placebo | – | 26.1%* | 30.1%* |
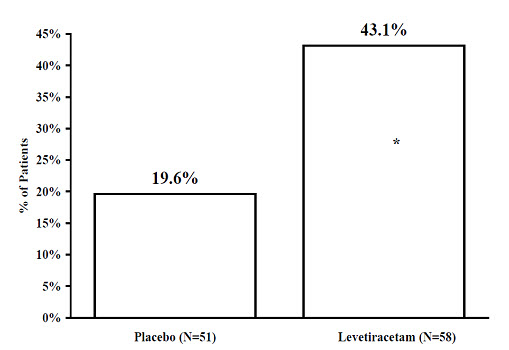
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the three treatment groups (x-axis) is presented in Figure 1.
Figure 1: Responder Rate (≥50% Reduction from Baseline) in Study 1
 |
* statistically significant versus placebo
Study 2
Study 2 was a double-blind, placebo-controlled, crossover study conducted at 62 centers in Europe comparing levetiracetam 1,000 mg/day (N=106), levetiracetam 2,000 mg/day (N=105), and placebo (N=111) given in equally divided doses twice daily.
The first period of the study (Period A) was designed to be analyzed as a parallel-group study. After a prospective baseline period of up to 12 weeks, patients were randomized to one of the three treatment groups described above. The 16-week treatment period consisted of the 4-week titration period followed by a 12-week fixed dose evaluation period, during which concomitant AED regimens were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). The results of the analysis of Period A are displayed in Table 11.
| Placebo (N=111) | Levetiracetam 1,000 mg/day (N=106) | Levetiracetam 2,000 mg/day (N=105) | |
|---|---|---|---|
| |||
Percent reduction in partial seizure frequency over placebo | – | 17.1%* | 21.4%* |
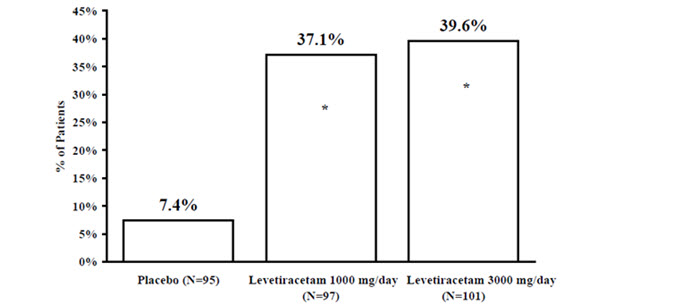
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the three treatment groups (x-axis) is presented in Figure 2.
Figure 2: Responder Rate (≥50% Reduction from Baseline) in Study 2: Period A
 |
* statistically significant versus placebo
The comparison of levetiracetam 2,000 mg/day to levetiracetam 1,000 mg/day for responder rate was statistically significant (P=0.02). Analysis of the trial as a cross-over yielded similar results.
Study 3
Study 3 was a double-blind, placebo-controlled, parallel-group study conducted at 47 centers in Europe comparing levetiracetam 3,000 mg/day (N=180) and placebo (N=104) in patients with refractory partial-onset seizures, with or without secondary generalization, receiving only one concomitant AED. Study drug was given in two divided doses. After a prospective baseline period of 12 weeks, patients were randomized to one of two treatment groups described above. The 16-week treatment period consisted of a 4-week titration period, followed by a 12-week fixed dose evaluation period, during which concomitant AED doses were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). Table 12 displays the results of the analysis of Study 3.
| Placebo (N=104) | Levetiracetam 3,000 mg/day (N=180) | |
|---|---|---|
| ||
Percent reduction in partial seizure frequency over placebo | – | 23.0%* |
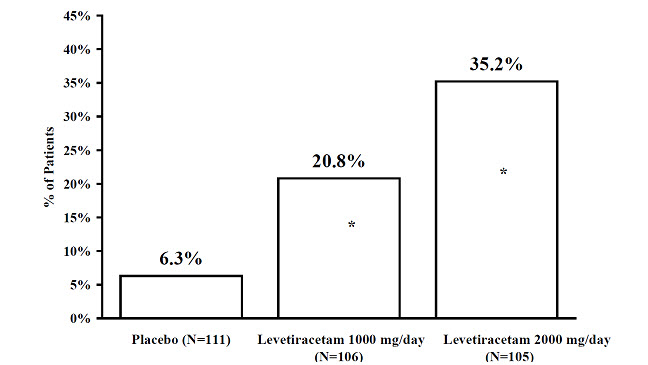
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 3.
Figure 3: Responder Rate (≥50% Reduction from Baseline) in Study 3
 |
* statistically significant versus placebo
Effectiveness in Partial-Onset Seizures in Pediatric Patients 4 Years to 16 Years of Age
Study 4 was a multicenter, randomized double-blind, placebo-controlled study, in pediatric patients 4 to 16 years of age with partial seizures uncontrolled by standard antiepileptic drugs (AEDs). Study 4 was conducted at 60 sites in North America. The study consisted of an 8-week baseline period and 4-week titration period followed by a 10-week evaluation period. Eligible patients who still experienced, on a stable dose of 1–2 AEDs, at least 4 partial-onset seizures during the 4 weeks prior to screening, as well as at least 4 partial-onset seizures in each of the two 4-week baseline periods, were randomized to receive either levetiracetam or placebo. Dosing was initiated at a dose of 20 mg/kg/day in two divided doses. During the treatment period, levetiracetam doses were adjusted in 20 mg/kg/day increments, at 2-week intervals to the target dose of 60 mg/kg/day. The primary measure of efficacy was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire 14-week randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥ 50% reduction from baseline in partial-onset seizure frequency per week). The enrolled population included 198 patients (levetiracetam N=101, placebo N=97) with refractory partial-onset seizures, whether or not secondarily generalized. Table 13 displays the results of Study 4.
| Placebo (N=97) | Levetiracetam (N=101) | |
|---|---|---|
| ||
Percent reduction in partial seizure frequency over placebo | – | 26.8%* |
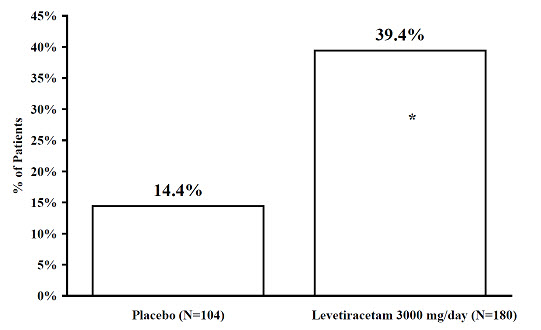
The percentage of patients (y-axis) who achieved ≥ 50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 4.
Figure 4: Responder Rate (≥ 50% Reduction from Baseline) in Study 4
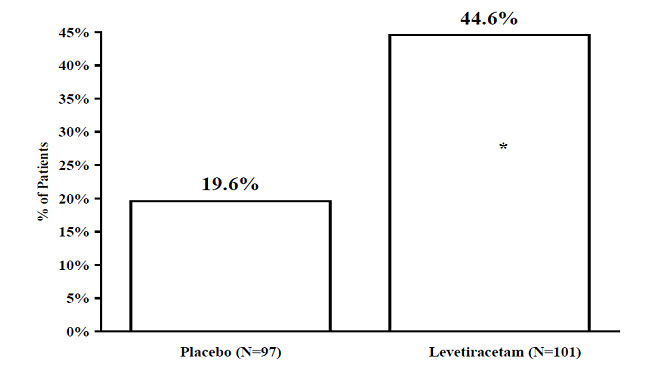 |
* statistically significant versus placebo
Effectiveness in Partial-Onset Seizures in Pediatric Patients 1 Month to < 4 Years of Age
Study 5 was a multicenter, randomized double-blind, placebo-controlled study, in pediatric patients 1 month to less than 4 years of age with partial seizures, uncontrolled by standard epileptic drugs (AEDs). Study 5 was conducted at 62 sites in North America, South America, and Europe. Study 5 consisted of a 5-day evaluation period, which included a 1-day titration period followed by a 4-day maintenance period. Eligible patients who experienced, on a stable dose of 1–2 AEDs, at least 2 partial-onset seizures during the 48-hour baseline video EEG were randomized to receive either levetiracetam or placebo. Randomization was stratified by age range as follows: 1 month to less than 6 months of age (N=4 treated with levetiracetam), 6 months to less than 1 year of age (N=8 treated with levetiracetam), 1 year to less than 2 years of age (N=20 treated with levetiracetam), and 2 years to less than 4 years of age (N=28 treated with levetiracetam). Levetiracetam dosing was determined by age and weight as follows: children 1 month to less than 6 months old were randomized to a target dose of 40 mg/kg/day, and children 6 months to less than 4 years old were randomized to a target dose of 50 mg/kg/day. The primary measure of efficacy was the responder rate (percent of patients with ≥ 50% reduction from baseline in average daily partial-onset seizure frequency) assessed by a blinded central reader using a 48-hour video EEG performed during the last two days of the 4-day maintenance period. The enrolled population included 116 patients (levetiracetam N=60, placebo N=56) with refractory partial-onset seizures, whether or not secondarily generalized. A total of 109 patients were included in the efficacy analysis. A statistically significant difference between levetiracetam and placebo was observed in Study 5 (see Figure 5). The treatment effect associated with levetiracetam was consistent across age groups.
Figure 5: Responder Rate for All Patients Ages 1 Month to < 4 Years (≥ 50% Reduction from Baseline) in Study 5
* statistically significant versus placebo
14.2 Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy
The effectiveness of levetiracetam as adjunctive therapy in patients 12 years of age and older with juvenile myoclonic epilepsy (JME) experiencing myoclonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (study 6), conducted at 37 sites in 14 countries. Eligible patients on a stable dose of 1 antiepileptic drug (AED) experiencing one or more myoclonic seizures per day for at least 8 days during the prospective 8-week baseline period were randomized to either levetiracetam or placebo (levetiracetam N=60, placebo N=60). Patients were titrated over 4 weeks to a target dose of 3,000 mg/day and treated at a stable dose of 3,000 mg/day over 12 weeks (evaluation period). Study drug was given in 2 divided doses. The primary measure of efficacy was the proportion of patients with at least 50% reduction in the number of days per week with one or more myoclonic seizures during the treatment period (titration + evaluation periods) as compared to baseline. Table 14 displays the results for the 113 patients with JME in this study.
| Placebo (N=59) | Levetiracetam (N=54) | |
|---|---|---|
| ||
Percentage of responders | 23.7% | 60.4%* |
14.3 Primary Generalized Tonic-Clonic Seizures
The effectiveness of levetiracetam as adjunctive therapy in patients 6 years of age and older with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic (PGTC) seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (study 7), conducted at 50 sites in 8 countries. Eligible patients on a stable dose of 1 or 2 antiepileptic drugs (AEDs) experiencing at least 3 PGTC seizures during the 8-week combined baseline period (at least one PGTC seizure during the 4 weeks prior to the prospective baseline period and at least one PGTC seizure during the 4-week prospective baseline period) were randomized to either levetiracetam or placebo. The 8-week combined baseline period is referred to as "baseline" in the remainder of this section. Patients were titrated over 4 weeks to a target dose of 3,000 mg/day for adults or a pediatric target dose of 60 mg/kg/day and treated at a stable dose of 3,000 mg/day (or 60 mg/kg/day for children) over 20 weeks (evaluation period). Study drug was given in 2 equally divided doses per day. The primary measure of efficacy was the percent reduction from baseline in weekly PGTC seizure frequency for levetiracetam and placebo treatment groups over the treatment period (titration + evaluation periods). The population included 164 patients (levetiracetam N=80, placebo N=84) with idiopathic generalized epilepsy (predominately juvenile myoclonic epilepsy, juvenile absence epilepsy, childhood absence epilepsy, or epilepsy with Grand Mal seizures on awakening) experiencing primary generalized tonic-clonic seizures. Each of these syndromes of idiopathic generalized epilepsy was well represented in this patient population.
There was a statistically significant decrease from baseline in PGTC frequency in the levetiracetam-treated patients compared to the placebo-treated patients in Study 7 (see Table 15).
| Placebo (N=84) | Levetiracetam (N=78) | |
|---|---|---|
| ||
Percentage reduction in PGTC seizure frequency | 44.6% | 77.6%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in PGTC seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 6.
Figure 6: Responder Rate (≥50% Reduction from Baseline) in PGTC Seizure Frequency per Week in Study 7
 |
* statistically significant versus placebo
Find Levetiracetam Injection medical information:
Find Levetiracetam Injection medical information:
Levetiracetam Injection Quick Finder
Health Professional Information
Clinical Studies
14 CLINICAL STUDIES
All clinical studies supporting the efficacy of levetiracetam utilized oral formulations. The finding of efficacy of levetiracetam injection is based on the results of studies using an oral formulation of levetiracetam, and on the demonstration of comparable bioavailability of the oral and parenteral formulations [see Clinical Pharmacology (12.3)].
14.1 Partial-Onset Seizures
Effectiveness in Partial-Onset Seizures in Adults
The effectiveness of levetiracetam for the treatment of partial-onset seizures in adults was established in three multicenter, randomized, double-blind, placebo-controlled clinical studies in patients who had refractory partial-onset seizures with or without secondary generalization. The tablet formulation was used in all these studies. In these studies, 904 patients were randomized to placebo, 1,000 mg, 2,000 mg, or 3,000 mg/day. Patients enrolled in Study 1 or Study 2 had refractory partial-onset seizures for at least two years and had taken two or more classical AEDs. Patients enrolled in Study 3 had refractory partial-onset seizures for at least 1 year and had taken one classical AED. At the time of the study, patients were taking a stable dose regimen of at least one and could take a maximum of two AEDs. During the baseline period, patients had to have experienced at least two partial-onset seizures during each 4-week period.
Study 1
Study 1 was a double-blind, placebo-controlled, parallel-group study conducted at 41 sites in the United States comparing levetiracetam 1,000 mg/day (N=97), levetiracetam 3,000 mg/day (N=101), and placebo (N=95) given in equally divided doses twice daily. After a prospective baseline period of 12 weeks, patients were randomized to one of the three treatment groups described above. The 18-week treatment period consisted of a 6-week titration period, followed by a 12-week fixed dose evaluation period, during which concomitant AED regimens were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). The results of the analysis of Study 1 are displayed in Table 10.
| Placebo (N=95) | Levetiracetam 1,000 mg/day (N=97) | Levetiracetam 3,000 mg/day (N=101) | |
|---|---|---|---|
| |||
Percent reduction in partial seizure frequency over placebo | – | 26.1%* | 30.1%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the three treatment groups (x-axis) is presented in Figure 1.
Figure 1: Responder Rate (≥50% Reduction from Baseline) in Study 1
 |
* statistically significant versus placebo
Study 2
Study 2 was a double-blind, placebo-controlled, crossover study conducted at 62 centers in Europe comparing levetiracetam 1,000 mg/day (N=106), levetiracetam 2,000 mg/day (N=105), and placebo (N=111) given in equally divided doses twice daily.
The first period of the study (Period A) was designed to be analyzed as a parallel-group study. After a prospective baseline period of up to 12 weeks, patients were randomized to one of the three treatment groups described above. The 16-week treatment period consisted of the 4-week titration period followed by a 12-week fixed dose evaluation period, during which concomitant AED regimens were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). The results of the analysis of Period A are displayed in Table 11.
| Placebo (N=111) | Levetiracetam 1,000 mg/day (N=106) | Levetiracetam 2,000 mg/day (N=105) | |
|---|---|---|---|
| |||
Percent reduction in partial seizure frequency over placebo | – | 17.1%* | 21.4%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the three treatment groups (x-axis) is presented in Figure 2.
Figure 2: Responder Rate (≥50% Reduction from Baseline) in Study 2: Period A
 |
* statistically significant versus placebo
The comparison of levetiracetam 2,000 mg/day to levetiracetam 1,000 mg/day for responder rate was statistically significant (P=0.02). Analysis of the trial as a cross-over yielded similar results.
Study 3
Study 3 was a double-blind, placebo-controlled, parallel-group study conducted at 47 centers in Europe comparing levetiracetam 3,000 mg/day (N=180) and placebo (N=104) in patients with refractory partial-onset seizures, with or without secondary generalization, receiving only one concomitant AED. Study drug was given in two divided doses. After a prospective baseline period of 12 weeks, patients were randomized to one of two treatment groups described above. The 16-week treatment period consisted of a 4-week titration period, followed by a 12-week fixed dose evaluation period, during which concomitant AED doses were held constant. The primary measure of effectiveness was a between group comparison of the percent reduction in weekly seizure frequency relative to placebo over the entire randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥50% reduction from baseline in partial-onset seizure frequency). Table 12 displays the results of the analysis of Study 3.
| Placebo (N=104) | Levetiracetam 3,000 mg/day (N=180) | |
|---|---|---|
| ||
Percent reduction in partial seizure frequency over placebo | – | 23.0%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 3.
Figure 3: Responder Rate (≥50% Reduction from Baseline) in Study 3
 |
* statistically significant versus placebo
Effectiveness in Partial-Onset Seizures in Pediatric Patients 4 Years to 16 Years of Age
Study 4 was a multicenter, randomized double-blind, placebo-controlled study, in pediatric patients 4 to 16 years of age with partial seizures uncontrolled by standard antiepileptic drugs (AEDs). Study 4 was conducted at 60 sites in North America. The study consisted of an 8-week baseline period and 4-week titration period followed by a 10-week evaluation period. Eligible patients who still experienced, on a stable dose of 1–2 AEDs, at least 4 partial-onset seizures during the 4 weeks prior to screening, as well as at least 4 partial-onset seizures in each of the two 4-week baseline periods, were randomized to receive either levetiracetam or placebo. Dosing was initiated at a dose of 20 mg/kg/day in two divided doses. During the treatment period, levetiracetam doses were adjusted in 20 mg/kg/day increments, at 2-week intervals to the target dose of 60 mg/kg/day. The primary measure of efficacy was a between group comparison of the percent reduction in weekly partial seizure frequency relative to placebo over the entire 14-week randomized treatment period (titration + evaluation period). Secondary outcome variables included the responder rate (incidence of patients with ≥ 50% reduction from baseline in partial-onset seizure frequency per week). The enrolled population included 198 patients (levetiracetam N=101, placebo N=97) with refractory partial-onset seizures, whether or not secondarily generalized. Table 13 displays the results of Study 4.
| Placebo (N=97) | Levetiracetam (N=101) | |
|---|---|---|
| ||
Percent reduction in partial seizure frequency over placebo | – | 26.8%* |
The percentage of patients (y-axis) who achieved ≥ 50% reduction in weekly seizure rates from baseline in partial-onset seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 4.
Figure 4: Responder Rate (≥ 50% Reduction from Baseline) in Study 4
 |
* statistically significant versus placebo
Effectiveness in Partial-Onset Seizures in Pediatric Patients 1 Month to < 4 Years of Age
Study 5 was a multicenter, randomized double-blind, placebo-controlled study, in pediatric patients 1 month to less than 4 years of age with partial seizures, uncontrolled by standard epileptic drugs (AEDs). Study 5 was conducted at 62 sites in North America, South America, and Europe. Study 5 consisted of a 5-day evaluation period, which included a 1-day titration period followed by a 4-day maintenance period. Eligible patients who experienced, on a stable dose of 1–2 AEDs, at least 2 partial-onset seizures during the 48-hour baseline video EEG were randomized to receive either levetiracetam or placebo. Randomization was stratified by age range as follows: 1 month to less than 6 months of age (N=4 treated with levetiracetam), 6 months to less than 1 year of age (N=8 treated with levetiracetam), 1 year to less than 2 years of age (N=20 treated with levetiracetam), and 2 years to less than 4 years of age (N=28 treated with levetiracetam). Levetiracetam dosing was determined by age and weight as follows: children 1 month to less than 6 months old were randomized to a target dose of 40 mg/kg/day, and children 6 months to less than 4 years old were randomized to a target dose of 50 mg/kg/day. The primary measure of efficacy was the responder rate (percent of patients with ≥ 50% reduction from baseline in average daily partial-onset seizure frequency) assessed by a blinded central reader using a 48-hour video EEG performed during the last two days of the 4-day maintenance period. The enrolled population included 116 patients (levetiracetam N=60, placebo N=56) with refractory partial-onset seizures, whether or not secondarily generalized. A total of 109 patients were included in the efficacy analysis. A statistically significant difference between levetiracetam and placebo was observed in Study 5 (see Figure 5). The treatment effect associated with levetiracetam was consistent across age groups.
Figure 5: Responder Rate for All Patients Ages 1 Month to < 4 Years (≥ 50% Reduction from Baseline) in Study 5
* statistically significant versus placebo
14.2 Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy
The effectiveness of levetiracetam as adjunctive therapy in patients 12 years of age and older with juvenile myoclonic epilepsy (JME) experiencing myoclonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (study 6), conducted at 37 sites in 14 countries. Eligible patients on a stable dose of 1 antiepileptic drug (AED) experiencing one or more myoclonic seizures per day for at least 8 days during the prospective 8-week baseline period were randomized to either levetiracetam or placebo (levetiracetam N=60, placebo N=60). Patients were titrated over 4 weeks to a target dose of 3,000 mg/day and treated at a stable dose of 3,000 mg/day over 12 weeks (evaluation period). Study drug was given in 2 divided doses. The primary measure of efficacy was the proportion of patients with at least 50% reduction in the number of days per week with one or more myoclonic seizures during the treatment period (titration + evaluation periods) as compared to baseline. Table 14 displays the results for the 113 patients with JME in this study.
| Placebo (N=59) | Levetiracetam (N=54) | |
|---|---|---|
| ||
Percentage of responders | 23.7% | 60.4%* |
14.3 Primary Generalized Tonic-Clonic Seizures
The effectiveness of levetiracetam as adjunctive therapy in patients 6 years of age and older with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic (PGTC) seizures was established in one multicenter, randomized, double-blind, placebo-controlled study (study 7), conducted at 50 sites in 8 countries. Eligible patients on a stable dose of 1 or 2 antiepileptic drugs (AEDs) experiencing at least 3 PGTC seizures during the 8-week combined baseline period (at least one PGTC seizure during the 4 weeks prior to the prospective baseline period and at least one PGTC seizure during the 4-week prospective baseline period) were randomized to either levetiracetam or placebo. The 8-week combined baseline period is referred to as "baseline" in the remainder of this section. Patients were titrated over 4 weeks to a target dose of 3,000 mg/day for adults or a pediatric target dose of 60 mg/kg/day and treated at a stable dose of 3,000 mg/day (or 60 mg/kg/day for children) over 20 weeks (evaluation period). Study drug was given in 2 equally divided doses per day. The primary measure of efficacy was the percent reduction from baseline in weekly PGTC seizure frequency for levetiracetam and placebo treatment groups over the treatment period (titration + evaluation periods). The population included 164 patients (levetiracetam N=80, placebo N=84) with idiopathic generalized epilepsy (predominately juvenile myoclonic epilepsy, juvenile absence epilepsy, childhood absence epilepsy, or epilepsy with Grand Mal seizures on awakening) experiencing primary generalized tonic-clonic seizures. Each of these syndromes of idiopathic generalized epilepsy was well represented in this patient population.
There was a statistically significant decrease from baseline in PGTC frequency in the levetiracetam-treated patients compared to the placebo-treated patients in Study 7 (see Table 15).
| Placebo (N=84) | Levetiracetam (N=78) | |
|---|---|---|
| ||
Percentage reduction in PGTC seizure frequency | 44.6% | 77.6%* |
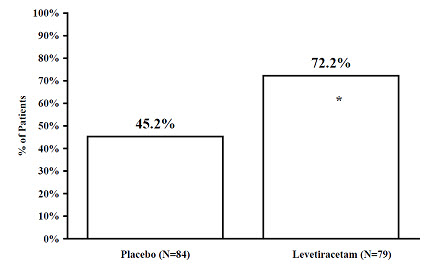
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in PGTC seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 6.
Figure 6: Responder Rate (≥50% Reduction from Baseline) in PGTC Seizure Frequency per Week in Study 7
 |
* statistically significant versus placebo
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5Pm ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.