dopamine Hydrochloride in 5% dextrose injection, USP Description
11 DESCRIPTION
Dopamine Hydrochloride in 5% Dextrose Injection, USP is a sterile, nonpyrogenic, premixed solution of dopamine hydrochloride in 5% dextrose injection for intravenous infusion.
Each 100 mL contains 80 mg (800 mcg/mL), 160 mg (1600 mcg/mL) or 320 mg (3200 mcg/mL) of dopamine HCl; 5 grams of hydrous dextrose, in Water for Injection, and 50 mg of sodium metabisulfite (a stabilizer); pH = 3.8 (2.5 to 4.5), and the following osmolar concentrations: 261, 269, or 286 mOsmol/liter, respectively. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment.
Dopamine HCl is chemically designated 3, 4-dihydroxyphenethylamine hydrochloride (C8H11NO2 ∙ HCl), a white crystalline powder freely soluble in water. Dopamine HCl has a molecular weight of 189.64 and it has the following structural formula:
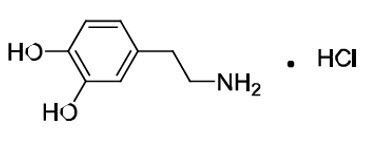
Dopamine (also referred to as 3-hydroxytyramine) is a naturally occurring endogenous catecholamine.
Dextrose, USP is chemically designated D-glucose monohydrate (C6H12O6 ∙ H2O), a hexose sugar freely soluble in water. The molecular weight of dextrose (D-glucose) monohydrate is 198.17 and it has the following structural formula:
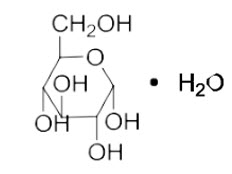
Water for Injection, USP is chemically designated H2O.
Find dopamine Hydrochloride in 5% dextrose injection, USP medical information:
Find dopamine Hydrochloride in 5% dextrose injection, USP medical information:
dopamine Hydrochloride in 5% dextrose injection, USP Quick Finder
Health Professional Information
Description
11 DESCRIPTION
Dopamine Hydrochloride in 5% Dextrose Injection, USP is a sterile, nonpyrogenic, premixed solution of dopamine hydrochloride in 5% dextrose injection for intravenous infusion.
Each 100 mL contains 80 mg (800 mcg/mL), 160 mg (1600 mcg/mL) or 320 mg (3200 mcg/mL) of dopamine HCl; 5 grams of hydrous dextrose, in Water for Injection, and 50 mg of sodium metabisulfite (a stabilizer); pH = 3.8 (2.5 to 4.5), and the following osmolar concentrations: 261, 269, or 286 mOsmol/liter, respectively. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment.
Dopamine HCl is chemically designated 3, 4-dihydroxyphenethylamine hydrochloride (C8H11NO2 ∙ HCl), a white crystalline powder freely soluble in water. Dopamine HCl has a molecular weight of 189.64 and it has the following structural formula:

Dopamine (also referred to as 3-hydroxytyramine) is a naturally occurring endogenous catecholamine.
Dextrose, USP is chemically designated D-glucose monohydrate (C6H12O6 ∙ H2O), a hexose sugar freely soluble in water. The molecular weight of dextrose (D-glucose) monohydrate is 198.17 and it has the following structural formula:

Water for Injection, USP is chemically designated H2O.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.