DAURISMO™ Clinical Studies
(glasdegib)
14 CLINICAL STUDIES
The efficacy of DAURISMO in combination with low-dose cytarabine was evaluated in a multicenter, open-label, randomized study (Study BRIGHT AML 1003, NCT01546038) that included 115 patients age 55 years or older with newly-diagnosed AML who met at least one of the following criteria: a) age ≥75 years, b) severe cardiac disease, c) baseline Eastern Cooperative Oncology Group (ECOG) performance status of 2, or d) baseline serum creatinine >1.3 mg/dL. Patients were randomized 2:1 to receive DAURISMO at a 100 mg daily dose with low-dose cytarabine 20 mg subcutaneously twice daily on days 1 to 10 of a 28-day cycle (N=77) or low-dose cytarabine alone (N=38) in 28-day cycles until disease progression or unacceptable toxicity. Patients were stratified by cytogenetic risk (good/intermediate or poor).
The baseline demographic and disease characteristics are shown in Table 6. The two treatment arms were generally balanced with respect to the baseline demographics and disease characteristics (see Table 6).
| Demographic and Disease Characteristics | DAURISMO With Low-Dose Cytarabine (N=77) | Low-Dose Cytarabine Alone (N=38) |
|---|---|---|
| Abbreviations: AML = acute myeloid leukemia; N = number of patients; ECOG PS = Eastern Cooperative Oncology Group Performance Status. | ||
| ||
Demographics | ||
Age | ||
Median (Min, Max) (Years) | 77 (64, 92) | 76 (58, 83) |
≥ 75 years N (%) | 47 (61) | 23 (61) |
Sex, N (%) | ||
Male | 59 (77) | 23 (61) |
Female | 18 (23) | 15 (39) |
Race, N (%) | ||
White | 75 (97) | 38 (100) |
Black or African American | 1 (1) | 0 (0) |
Asian | 1 (1) | 0 (0) |
Disease History, N (%) | ||
De Novo AML | 38 (49) | 18 (47) |
Secondary AML | 39 (51) | 20 (53) |
Prior Hypomethylating Agent Use | 11 (14) | 6 (16) |
ECOG PS*, N (%) | ||
0 to 1 | 35 (46) | 20 (53) |
2 | 41 (53) | 18 (47) |
Cytogenetic Risk Status, N (%) | ||
Good/Intermediate | 48 (62) | 21 (55) |
Poor | 29 (38) | 17 (45) |
Baseline Severe Cardiac Disease | 51 (66) | 20 (53) |
Baseline Serum Creatinine >1.3 mg/dL | 15 (19) | 5 (13) |
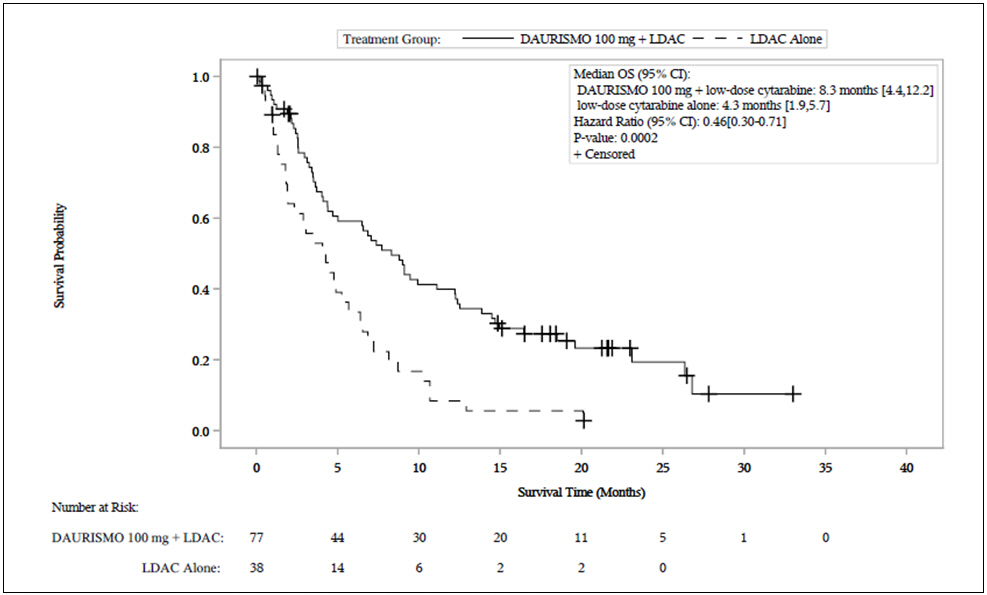
Efficacy was established on the basis of overall survival (OS) from the date of randomization to death from any cause. With a median follow-up of approximately 20 months, the DAURISMO with low-dose cytarabine arm was superior to low-dose cytarabine alone arm (Figure 1). The efficacy results are shown in Table 7. Improvement in OS was consistent across prespecified cytogenetic risk subgroups.
| Endpoint/Study Population | DAURISMO With Low-Dose Cytarabine | Low-Dose Cytarabine Alone |
|---|---|---|
| Abbreviations: AML = acute myeloid leukemia; N = number of patients; OS = overall survival; CI = confidence interval; CR = complete response. | ||
OS | N=77 | N=38 |
Median survival, months (95% CI) | 8.3 (4.4, 12.2) | 4.3 (1.9, 5.7) |
Hazard ratio (95% CI)* | 0.46 (0.30, 0.71) | |
p-value† | 0.0002 | |
CR | N=14 | N=1 |
CR rate (in %, 95% CI) | 18.2 (10.3, 28.6) | 2.6 (0.1, 13.8) |
Figure 1. BRIGHT AML 1003 – Kaplan-Meier Plot of Overall Survival for Patients with AML
Abbreviations: CI = confidence interval; OS = overall survival; LDAC = low-dose cytarabine.
Find DAURISMO™ medical information:
Find DAURISMO™ medical information:
DAURISMO™ Quick Finder
Health Professional Information
Clinical Studies
14 CLINICAL STUDIES
The efficacy of DAURISMO in combination with low-dose cytarabine was evaluated in a multicenter, open-label, randomized study (Study BRIGHT AML 1003, NCT01546038) that included 115 patients age 55 years or older with newly-diagnosed AML who met at least one of the following criteria: a) age ≥75 years, b) severe cardiac disease, c) baseline Eastern Cooperative Oncology Group (ECOG) performance status of 2, or d) baseline serum creatinine >1.3 mg/dL. Patients were randomized 2:1 to receive DAURISMO at a 100 mg daily dose with low-dose cytarabine 20 mg subcutaneously twice daily on days 1 to 10 of a 28-day cycle (N=77) or low-dose cytarabine alone (N=38) in 28-day cycles until disease progression or unacceptable toxicity. Patients were stratified by cytogenetic risk (good/intermediate or poor).
The baseline demographic and disease characteristics are shown in Table 6. The two treatment arms were generally balanced with respect to the baseline demographics and disease characteristics (see Table 6).
| Demographic and Disease Characteristics | DAURISMO With Low-Dose Cytarabine (N=77) | Low-Dose Cytarabine Alone (N=38) |
|---|---|---|
| Abbreviations: AML = acute myeloid leukemia; N = number of patients; ECOG PS = Eastern Cooperative Oncology Group Performance Status. | ||
| ||
Demographics | ||
Age | ||
Median (Min, Max) (Years) | 77 (64, 92) | 76 (58, 83) |
≥ 75 years N (%) | 47 (61) | 23 (61) |
Sex, N (%) | ||
Male | 59 (77) | 23 (61) |
Female | 18 (23) | 15 (39) |
Race, N (%) | ||
White | 75 (97) | 38 (100) |
Black or African American | 1 (1) | 0 (0) |
Asian | 1 (1) | 0 (0) |
Disease History, N (%) | ||
De Novo AML | 38 (49) | 18 (47) |
Secondary AML | 39 (51) | 20 (53) |
Prior Hypomethylating Agent Use | 11 (14) | 6 (16) |
ECOG PS*, N (%) | ||
0 to 1 | 35 (46) | 20 (53) |
2 | 41 (53) | 18 (47) |
Cytogenetic Risk Status, N (%) | ||
Good/Intermediate | 48 (62) | 21 (55) |
Poor | 29 (38) | 17 (45) |
Baseline Severe Cardiac Disease | 51 (66) | 20 (53) |
Baseline Serum Creatinine >1.3 mg/dL | 15 (19) | 5 (13) |
Efficacy was established on the basis of overall survival (OS) from the date of randomization to death from any cause. With a median follow-up of approximately 20 months, the DAURISMO with low-dose cytarabine arm was superior to low-dose cytarabine alone arm (Figure 1). The efficacy results are shown in Table 7. Improvement in OS was consistent across prespecified cytogenetic risk subgroups.
| Endpoint/Study Population | DAURISMO With Low-Dose Cytarabine | Low-Dose Cytarabine Alone |
|---|---|---|
| Abbreviations: AML = acute myeloid leukemia; N = number of patients; OS = overall survival; CI = confidence interval; CR = complete response. | ||
OS | N=77 | N=38 |
Median survival, months (95% CI) | 8.3 (4.4, 12.2) | 4.3 (1.9, 5.7) |
Hazard ratio (95% CI)* | 0.46 (0.30, 0.71) | |
p-value† | 0.0002 | |
CR | N=14 | N=1 |
CR rate (in %, 95% CI) | 18.2 (10.3, 28.6) | 2.6 (0.1, 13.8) |
Figure 1. BRIGHT AML 1003 – Kaplan-Meier Plot of Overall Survival for Patients with AML
Abbreviations: CI = confidence interval; OS = overall survival; LDAC = low-dose cytarabine.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.