CAVERJECT® IMPULSE Dosage and Administration
(alprostadil)
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
- •
- Alprostadil is available in different strengths and presentations. Determine the most suitable dose and presentation for each patient. Use a new syringe for each dose of CAVERJECT.
- •
- Titrate the dose of CAVERJECT IMPULSE for each patient to the lowest effective dose.
- •
- CAVERJECT IMPULSE doses greater than 60 mcg are not recommended.
- •
- Administer the first doses of CAVERJECT IMPULSE in the health care provider’s office by medically trained personnel.
- •
- Instruct the patient on proper use and assess that they are well trained in the self-injection technique prior to initiation of home use. Refer to the Patient Information and Instructions for Use.
- •
- Re-evaluate patients regularly (every 3 months or as clinically appropriate) and determine if dosage adjustments are needed.
2.2 Recommended Dosage for Erectile Dysfunction
Erectile Dysfunction of Vasculogenic, Psychogenic, or Mixed Etiology.
- •
- Initiate dosing with 2.5 mcg of CAVERJECT IMPULSE intracavernousally as recommended [see Dosage and Administration 2.4].
- •
- If there is a partial response at 2.5 mcg, administer another dose of 2.5 mcg within 1 hour.
- •
- During titration, no more than 2 doses should be given within a 24-hour period.
- •
- If additional titration is required, administer doses in increments of 5 to 10 mcg at least 24 hours apart.
- •
- The optimal dose should produce an erection suitable for intercourse that does not exceed a duration of 1 hour.
- •
- The patient must stay in the health care provider’s office until complete detumescence occurs.
Repeat the titration as necessary until the optimal dose is achieved. Doses greater than 60 mcg are not recommended.
Erectile Dysfunction of Pure Neurogenic Etiology (Spinal Cord Injury):
- •
- Initiate dosing with 1.25 mcg of alprostadil using CAVERJECT.
- •
- If there is a partial response, administer another dose of CAVERJECT of 1.25 mcg within 1 hour.
- •
- No more than 2 doses during initial titration should be given within a 24-hour period.
- •
- If additional titration is required, administer a dose of 5 mcg at least 24 hours later.
- •
- The optimal dose should produce an erection suitable for intercourse that does not exceed a duration of 1 hour.
- •
- The patient must stay in the health care provider’s office until complete detumescence occurs.
Repeat the titration as necessary until the optimal dose is achieved. Doses greater than 60 mcg are not recommended.
Maintenance Dosage for Patient Home Use:
- •
- Once the dose of CAVERJECT IMPULSE has been determined in the health care provider’s office, additional dose adjustment may be required after consultation with the health care provider. Adjust the dose in accordance with the titration guidelines described above.
- •
- The recommended frequency of injection is no more than 3 times weekly, with at least 24 hours between each dose.
Adjunct to the Diagnosis of Erectile Dysfunction
To diagnose erectile dysfunction (pharmacologic testing), inject CAVERJECT IMPULSE intracavernosally and monitor patients for the occurrence of an erection. Extensions of this testing are the use of CAVERJECT as an adjunct to laboratory investigations, such as duplex or Doppler imaging. For any of these tests, use a single dose of CAVERJECT IMPULSE that induces a rigid erection. Use the dose regimen for ‘Erectile Dysfunction of Vasculogenic, Psychogenic, or Mixed Etiology’ above.
2.3 Syringe Preparation Instructions
- 1.
- Select the CAVERJECT IMPULSE syringe based upon dose to be administered.
| Syringe Strength | Reconstituted Concentration | Dosages Available for Delivery after Reconstitution | |||
|---|---|---|---|---|---|
10 mcg | 10 mcg/0.5 mL | 2.5 mcg | 5 mcg | 7.5 mcg | 10 mcg |
20 mcg | 20 mcg/0.5 mL | 5 mcg | 10 mcg | 15 mcg | 20 mcg |
- 2.
- Open the sealed plastic tray. Remove the syringe, the needle assembly, and the alcohol swabs from the tray. The syringe has a dose window and a plunger. The needle assembly is a sealed unit that contains the outer protective cap, the inner protective cap, and the superfine needle.
- 3.
- Use the alcohol swab to wipe the rubber membrane at the tip of the syringe. Pick up the needle assembly, grasp the paper tab, and peel off the paper cover (the lid).
- 4.
- Hold the needle assembly by the cap and press the needle assembly onto the tip of the syringe. Turn it clockwise until the needle assembly is firmly locked into place.
- 5.
- Remove the outer protective cap from the needle by twisting it clockwise. Do not yet remove the inner protective cap, the thin plastic tube that directly covers the needle.
- 6.
- Hold the syringe system with the needle pointing upward. The plunger rod should still be in the fully extended position, with all of the threads visible. Slowly rotate the plunger rod clockwise until it goes all the way in and stops. Do not push on the plunger while trying to rotate it.
- 7.
- Turn the syringe upside down several times to make sure the solution is evenly mixed. The solution should be clear. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. The product should not be used if particulate matter or discoloration are present.
- 8.
- Hold the syringe with the needle upward and carefully remove the inner protective cap from the needle. Lightly tap the glass cartridge a few times with your finger until any large bubbles disappear up into the tip. With the syringe pointed upward, push in the plunger rod until it stops to push any air out.
- 9.
- To set the dose: locate the dose window on the syringe and then slowly turn the plunger rod clockwise until the correct dose number appears in the center of the window. The syringe is now ready for use. If you pass the correct number, keep turning the plunger in the same direction until the correct number comes around again – do not try to turn it backward.
- 10.
- After reconstitution, the syringe should be used within 24 hours when stored between 36 to 77°F (2°C to 25°C). Do not freeze. CAVERJECT IMPULSE is for single use only. Discard the injection delivery system and any remaining solution after use.
2.4 Administration Instructions
- •
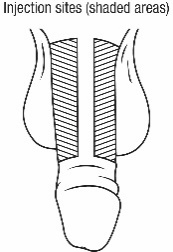
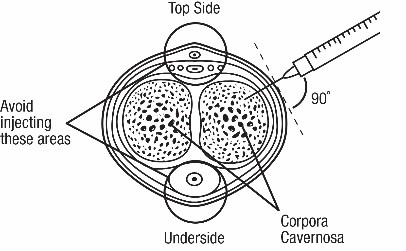
- Administer CAVERJECT IMPUSLE intracavernosally along the dorso-lateral aspect of the proximal third of the penis. See Figures A and B below.
Figure A
Figure B
- •
- Wipe the intended injection site with an alcohol swab prior to injection.
- •
- Avoid visible veins during injection.
- •
- Alternate the side of the penis that is injected and the site of injection.
- •
- Compress the site of injection with an alcohol swab or sterile gauze for 5 minutes.
- •
- Each CAVERJECT IMPULSE syringe is intended for single use only (one dose only) and should be discarded after use.
Find CAVERJECT® IMPULSE medical information:
Find CAVERJECT® IMPULSE medical information:
CAVERJECT® IMPULSE Quick Finder
Health Professional Information
Dosage and Administration
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
- •
- Alprostadil is available in different strengths and presentations. Determine the most suitable dose and presentation for each patient. Use a new syringe for each dose of CAVERJECT.
- •
- Titrate the dose of CAVERJECT IMPULSE for each patient to the lowest effective dose.
- •
- CAVERJECT IMPULSE doses greater than 60 mcg are not recommended.
- •
- Administer the first doses of CAVERJECT IMPULSE in the health care provider’s office by medically trained personnel.
- •
- Instruct the patient on proper use and assess that they are well trained in the self-injection technique prior to initiation of home use. Refer to the Patient Information and Instructions for Use.
- •
- Re-evaluate patients regularly (every 3 months or as clinically appropriate) and determine if dosage adjustments are needed.
2.2 Recommended Dosage for Erectile Dysfunction
Erectile Dysfunction of Vasculogenic, Psychogenic, or Mixed Etiology.
- •
- Initiate dosing with 2.5 mcg of CAVERJECT IMPULSE intracavernousally as recommended [see Dosage and Administration 2.4].
- •
- If there is a partial response at 2.5 mcg, administer another dose of 2.5 mcg within 1 hour.
- •
- During titration, no more than 2 doses should be given within a 24-hour period.
- •
- If additional titration is required, administer doses in increments of 5 to 10 mcg at least 24 hours apart.
- •
- The optimal dose should produce an erection suitable for intercourse that does not exceed a duration of 1 hour.
- •
- The patient must stay in the health care provider’s office until complete detumescence occurs.
Repeat the titration as necessary until the optimal dose is achieved. Doses greater than 60 mcg are not recommended.
Erectile Dysfunction of Pure Neurogenic Etiology (Spinal Cord Injury):
- •
- Initiate dosing with 1.25 mcg of alprostadil using CAVERJECT.
- •
- If there is a partial response, administer another dose of CAVERJECT of 1.25 mcg within 1 hour.
- •
- No more than 2 doses during initial titration should be given within a 24-hour period.
- •
- If additional titration is required, administer a dose of 5 mcg at least 24 hours later.
- •
- The optimal dose should produce an erection suitable for intercourse that does not exceed a duration of 1 hour.
- •
- The patient must stay in the health care provider’s office until complete detumescence occurs.
Repeat the titration as necessary until the optimal dose is achieved. Doses greater than 60 mcg are not recommended.
Maintenance Dosage for Patient Home Use:
- •
- Once the dose of CAVERJECT IMPULSE has been determined in the health care provider’s office, additional dose adjustment may be required after consultation with the health care provider. Adjust the dose in accordance with the titration guidelines described above.
- •
- The recommended frequency of injection is no more than 3 times weekly, with at least 24 hours between each dose.
Adjunct to the Diagnosis of Erectile Dysfunction
To diagnose erectile dysfunction (pharmacologic testing), inject CAVERJECT IMPULSE intracavernosally and monitor patients for the occurrence of an erection. Extensions of this testing are the use of CAVERJECT as an adjunct to laboratory investigations, such as duplex or Doppler imaging. For any of these tests, use a single dose of CAVERJECT IMPULSE that induces a rigid erection. Use the dose regimen for ‘Erectile Dysfunction of Vasculogenic, Psychogenic, or Mixed Etiology’ above.
2.3 Syringe Preparation Instructions
- 1.
- Select the CAVERJECT IMPULSE syringe based upon dose to be administered.
| Syringe Strength | Reconstituted Concentration | Dosages Available for Delivery after Reconstitution | |||
|---|---|---|---|---|---|
10 mcg | 10 mcg/0.5 mL | 2.5 mcg | 5 mcg | 7.5 mcg | 10 mcg |
20 mcg | 20 mcg/0.5 mL | 5 mcg | 10 mcg | 15 mcg | 20 mcg |
- 2.
- Open the sealed plastic tray. Remove the syringe, the needle assembly, and the alcohol swabs from the tray. The syringe has a dose window and a plunger. The needle assembly is a sealed unit that contains the outer protective cap, the inner protective cap, and the superfine needle.
- 3.
- Use the alcohol swab to wipe the rubber membrane at the tip of the syringe. Pick up the needle assembly, grasp the paper tab, and peel off the paper cover (the lid).
- 4.
- Hold the needle assembly by the cap and press the needle assembly onto the tip of the syringe. Turn it clockwise until the needle assembly is firmly locked into place.
- 5.
- Remove the outer protective cap from the needle by twisting it clockwise. Do not yet remove the inner protective cap, the thin plastic tube that directly covers the needle.
- 6.
- Hold the syringe system with the needle pointing upward. The plunger rod should still be in the fully extended position, with all of the threads visible. Slowly rotate the plunger rod clockwise until it goes all the way in and stops. Do not push on the plunger while trying to rotate it.
- 7.
- Turn the syringe upside down several times to make sure the solution is evenly mixed. The solution should be clear. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. The product should not be used if particulate matter or discoloration are present.
- 8.
- Hold the syringe with the needle upward and carefully remove the inner protective cap from the needle. Lightly tap the glass cartridge a few times with your finger until any large bubbles disappear up into the tip. With the syringe pointed upward, push in the plunger rod until it stops to push any air out.
- 9.
- To set the dose: locate the dose window on the syringe and then slowly turn the plunger rod clockwise until the correct dose number appears in the center of the window. The syringe is now ready for use. If you pass the correct number, keep turning the plunger in the same direction until the correct number comes around again – do not try to turn it backward.
- 10.
- After reconstitution, the syringe should be used within 24 hours when stored between 36 to 77°F (2°C to 25°C). Do not freeze. CAVERJECT IMPULSE is for single use only. Discard the injection delivery system and any remaining solution after use.
2.4 Administration Instructions
- •
- Administer CAVERJECT IMPUSLE intracavernosally along the dorso-lateral aspect of the proximal third of the penis. See Figures A and B below.
Figure A
Figure B
- •
- Wipe the intended injection site with an alcohol swab prior to injection.
- •
- Avoid visible veins during injection.
- •
- Alternate the side of the penis that is injected and the site of injection.
- •
- Compress the site of injection with an alcohol swab or sterile gauze for 5 minutes.
- •
- Each CAVERJECT IMPULSE syringe is intended for single use only (one dose only) and should be discarded after use.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.