bupivacaine hydrochloride injection, USP - SPINAL Description
11 DESCRIPTION
BUPIVACAINE SPINAL (bupivacaine hydrochloride in dextrose injection) is an amide-local anesthetic and sterile hyperbaric aqueous solution. The route of administration for BUPIVACAINE SPINAL is by subarachnoid injection. BUPIVACAINE SPINAL contains bupivacaine hydrochloride, as the active pharmaceutical ingredient and also contains Dextrose, as baricity agent.
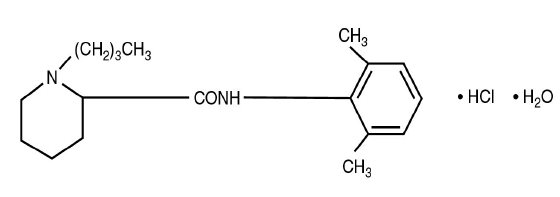
Bupivacaine Hydrochloride (monohydrate) chemical name is 2-piperidinecarboxamide, 1-butyl-N-(2,6-dimethylphenyl)-, monohydrochloride, monohydrate, a white crystalline powder that is freely soluble in 95 percent ethanol, soluble in water, and slightly soluble in chloroform or acetone. Bupivacaine Hydrochloride (monohydrate) has a molecular formula of C18H28N2O·HCl·H2O and molecular weight of 342.90 g/mol and has the following structural formula:
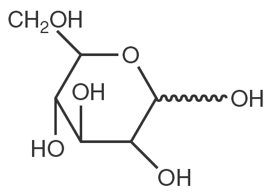
Dextrose chemical name is D-glucopyranose. Dextrose (anhydrous) has a molecular formula of C6H12O6, molecular weight of 180.16 g/mol and has the following structural formula:
BUPIVACAINE SPINAL (bupivacaine hydrochloride in dextrose injection) is a clear and colorless sterile hyperbaric solution.
Each mL of BUPIVACAINE SPINAL contains 7.5 mg bupivacaine hydrochloride (anhydrous) (equivalent to 7.9 mg of bupivacaine hydrochloride monohydrate), 82.5 mg dextrose (anhydrous) as baricity agent, and sodium hydroxide and hydrochloric acid as pH adjusters in water for injection.
BUPIVACAINE SPINAL pH is between 4.0 and 6.5.
The specific gravity of BUPIVACAINE SPINAL is between 1.030 and 1.035 at 25°C and 1.03 at 37°C.
BUPIVACAINE SPINAL does not contain any preservatives.
Find bupivacaine hydrochloride injection, USP - SPINAL medical information:
Find bupivacaine hydrochloride injection, USP - SPINAL medical information:
bupivacaine hydrochloride injection, USP - SPINAL Quick Finder
Health Professional Information
Description
11 DESCRIPTION
BUPIVACAINE SPINAL (bupivacaine hydrochloride in dextrose injection) is an amide-local anesthetic and sterile hyperbaric aqueous solution. The route of administration for BUPIVACAINE SPINAL is by subarachnoid injection. BUPIVACAINE SPINAL contains bupivacaine hydrochloride, as the active pharmaceutical ingredient and also contains Dextrose, as baricity agent.
Bupivacaine Hydrochloride (monohydrate) chemical name is 2-piperidinecarboxamide, 1-butyl-N-(2,6-dimethylphenyl)-, monohydrochloride, monohydrate, a white crystalline powder that is freely soluble in 95 percent ethanol, soluble in water, and slightly soluble in chloroform or acetone. Bupivacaine Hydrochloride (monohydrate) has a molecular formula of C18H28N2O·HCl·H2O and molecular weight of 342.90 g/mol and has the following structural formula:
Dextrose chemical name is D-glucopyranose. Dextrose (anhydrous) has a molecular formula of C6H12O6, molecular weight of 180.16 g/mol and has the following structural formula:
BUPIVACAINE SPINAL (bupivacaine hydrochloride in dextrose injection) is a clear and colorless sterile hyperbaric solution.
Each mL of BUPIVACAINE SPINAL contains 7.5 mg bupivacaine hydrochloride (anhydrous) (equivalent to 7.9 mg of bupivacaine hydrochloride monohydrate), 82.5 mg dextrose (anhydrous) as baricity agent, and sodium hydroxide and hydrochloric acid as pH adjusters in water for injection.
BUPIVACAINE SPINAL pH is between 4.0 and 6.5.
The specific gravity of BUPIVACAINE SPINAL is between 1.030 and 1.035 at 25°C and 1.03 at 37°C.
BUPIVACAINE SPINAL does not contain any preservatives.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.